Thermal activation and photoactivation of visual pigments
- PMID: 15189862
- PMCID: PMC1304267
- DOI: 10.1529/biophysj.103.035626
Thermal activation and photoactivation of visual pigments
Abstract
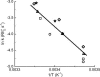
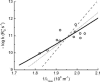
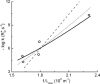
A visual pigment molecule in a retinal photoreceptor cell can be activated not only by absorption of a photon but also "spontaneously" by thermal energy. Current estimates of the activation energies for these two processes in vertebrate rod and cone pigments are on the order of 40-50 kcal/mol for activation by light and 20-25 kcal/mol for activation by heat, which has forced the conclusion that the two follow quite different molecular routes. It is shown here that the latter estimates, derived from the temperature dependence of the rate of pigment-initiated "dark events" in rods, depend on the unrealistic assumption that thermal activation of a complex molecule like rhodopsin (or even its 11-cis retinaldehyde chromophore) happens through a simple process, somewhat like the collision of gas molecules. When the internal energy present in the many vibrational modes of the molecule is taken into account, the thermal energy distribution of the molecules cannot be described by Boltzmann statistics, and conventional Arrhenius analysis gives incorrect estimates for the energy barrier. When the Boltzmann distribution is replaced by one derived by Hinshelwood for complex molecules with many vibrational modes, the same experimental data become consistent with thermal activation energies that are close to or even equal to the photoactivation energies. Thus activation by light and by heat may in fact follow the same molecular route, starting with 11-cis to all-trans isomerization of the chromophore in the native (resting) configuration of the opsin. Most importantly, the same model correctly predicts the empirical correlation between the wavelength of maximum absorbance and the rate of thermal activation in the whole set of visual pigments studied.
Figures
References
-
- Aho, A. C., K. Donner, C. Hyden, L. O. Larsen, and T. Reuter. 1988. Low retinal noise in animals with low body temperature allows high visual sensitivity. Nature. 334:348–350. - PubMed
-
- Ala-Laurila, P., R. Albert, P. Saarinen, A. Koskelainen, and K. Donner. 2003. The thermal contribution to photoactivation in A2 visual pigments studied by temperature effects on spectral properties. Vis. Neurosci. 20:411–419. - PubMed
-
- Ala-Laurila, P., J. Pahlberg, A. Koskelainen, and K. Donner. 2004. On the relation between the photoactivation energy and the absorbance spectrum of visual pigments. Vision Res. In press. - PubMed
-
- Ala-Laurila, P., P. Saarinen, R. Albert, A. Koskelainen, and K. Donner. 2002. Temperature effects on spectral properties of red and green rods in toad retina. Vis. Neurosci. 19:781–792. - PubMed
-
- Ashmore, J. F., and G. Falk. 1977. Dark noise in retinal bipolar cells and stability of rhodopsin in rods. Nature. 270:69–71. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

