Efficient production of germline transgenic chickens using lentiviral vectors
- PMID: 15192698
- PMCID: PMC1299092
- DOI: 10.1038/sj.embor.7400171
Efficient production of germline transgenic chickens using lentiviral vectors
Abstract
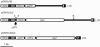
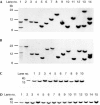
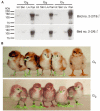
An effective method for genetic modification of chickens has yet to be developed. An efficient technology, enabling production of transgenic birds at high frequency and with reliable expression of transgenes, will have many applications, both in basic research and in biotechnology. We investigated the efficiency with which lentiviral vectors could transduce the chicken germ line and examined the expression of introduced reporter transgenes. Ten founder cockerels transmitted the vector to between 4% and 45% of their offspring and stable transmission to the G2 generation was demonstrated. Analysis of expression of reporter gene constructs in several transgenic lines showed a conserved expression profile between individuals that was maintained after transmission through the germ line. These data demonstrate that lentiviral vectors can be used to generate transgenic lines with an efficiency in the order of 100-fold higher than any previously published method, with no detectable silencing of transgene expression between generations.
Figures


References
-
- Bienemann A (2003) Long-term replacement of a mutated non-functional CNS gene: reversal of hypothalamic diabetes insipidus using an EIAV-based lentiviral vector expressing arginine vasopressin. Mol Ther 7: 588–596 - PubMed
-
- Bosselman RA et al. (1989) Germline transmission of exogenous genes in the chicken. Science 243: 533–535 - PubMed
-
- Brown WRA, Hubbard SJ, Tickle C, Wilson SA (2003) The chicken as a model for largescale analysis of vertebrate gene function. Nat Rev Genet 4: 87–98 - PubMed
-
- Corcoran J et al. (2002) Retinoic acid receptor β2 and neurite outgrowth in the adult mouse spinal cord in vitro. J Cell Sci 115: 3779–3786 - PubMed
-
- Eyal-Giladi H, Kochav S (1976) From cleavage to primitive streak formation: a complementary normal table and a new look at the first stages of development of the chick. Dev Biol 49: 321–327 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

