Time dependent risk of gastrointestinal complications induced by non-steroidal anti-inflammatory drug use: a consensus statement using a meta-analytic approach
- PMID: 15194568
- PMCID: PMC1755051
- DOI: 10.1136/ard.2003.015925
Time dependent risk of gastrointestinal complications induced by non-steroidal anti-inflammatory drug use: a consensus statement using a meta-analytic approach
Abstract
Objectives: To provide an updated document assessing the global, NSAID-specific, and time dependent risk of gastrointestinal (GI) complications through meta-analyses of high quality studies.
Methods: An exhaustive systematic search was performed. Inclusion criteria were: RCT or controlled study, duration of 5 days at least, inactive control, assessment of minor or major NSAID adverse effects, publication range January 1985 to January 2003. The publications retrieved were assessed during a specifically dedicated WHO meeting including leading experts in all related fields. Statistics were performed conservatively. Meta-regression was performed by regressing NSAID adjusted estimates against study duration categories.
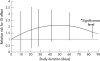
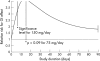
Results: Among RCT data, indolic derivates provided a significantly higher risk of GI complications related to NSAID use than for non-users: RR = 2.25 (1.00; 5.08) than did other compounds: naproxen: RR = 1.83 (1.25; 2.68); diclofenac: RR = 1.73 (1.21; 2.46); piroxicam: RR = 1.66 (1.14; 2.44); tenoxicam: RR = 1.43 (0.40; 5.14); meloxicam: RR = 1.24 (0.98; 1.56), and ibuprofen: RR = 1.19 (0.93; 1.54). Indometacin users had a maximum relative risk for complication at 14 days. The other compounds presented a better profile, with a maximum risk at 50 days. Significant additional risk factors included age, dose, and underlying disease. The controlled cohort studies provided higher estimates: RR = 2.22 (1.7; 2.9). Publication bias testing was significant, towards a selective publication of deleterious effects of NSAIDs from small sized studies.
Conclusion: This meta-analysis characterised the "compound" and "time" aspects of the GI toxicity of non-selective NSAIDs. The risk/benefit ratio of such compounds should thus be carefully and individually evaluated at the start of long term treatment.
Figures



Similar articles
-
Evaluation of risk profiles for gastrointestinal and cardiovascular adverse effects in nonselective NSAID and COX-2 inhibitor users: a cohort study using pharmacy dispensing data in The Netherlands.Drug Saf. 2008;31(2):143-58. doi: 10.2165/00002018-200831020-00004. Drug Saf. 2008. PMID: 18217790
-
Aspirin and other non-steroidal anti-inflammatory drugs for the prevention of dementia.Cochrane Database Syst Rev. 2020 Apr 30;4(4):CD011459. doi: 10.1002/14651858.CD011459.pub2. Cochrane Database Syst Rev. 2020. PMID: 32352165 Free PMC article.
-
Gastrointestinal tolerability of NSAIDs in elderly patients: a pooled analysis of 21 randomized clinical trials with celecoxib and nonselective NSAIDs.Curr Med Res Opin. 2011 Jul;27(7):1359-66. doi: 10.1185/03007995.2011.581274. Epub 2011 May 12. Curr Med Res Opin. 2011. PMID: 21561397 Review.
-
Relationship between diclofenac dose and risk of gastrointestinal and cardiovascular events: meta-regression based on two systematic literature reviews.Clin Ther. 2014 Jun 1;36(6):906-17. doi: 10.1016/j.clinthera.2014.04.012. Epub 2014 May 23. Clin Ther. 2014. PMID: 24863260
-
Gastrointestinal tolerability of meloxicam compared to diclofenac in osteoarthritis patients. International MELISSA Study Group. Meloxicam Large-scale International Study Safety Assessment.Br J Rheumatol. 1998 Sep;37(9):937-45. doi: 10.1093/rheumatology/37.9.937. Br J Rheumatol. 1998. PMID: 9783757 Clinical Trial.
Cited by
-
Adverse drug reactions and drug-drug interactions with over-the-counter NSAIDs.Ther Clin Risk Manag. 2015 Jul 15;11:1061-75. doi: 10.2147/TCRM.S79135. eCollection 2015. Ther Clin Risk Manag. 2015. PMID: 26203254 Free PMC article. Review.
-
Safety of the nonselective NSAID nabumetone : focus on gastrointestinal tolerability.Drug Saf. 2008;31(6):485-503. doi: 10.2165/00002018-200831060-00004. Drug Saf. 2008. PMID: 18484783 Review.
-
Rectal indomethacin and diclofenac are equally efficient in preventing pancreatitis following endoscopic retrograde cholangiopancreatography in average-risk patients.JGH Open. 2021 Aug 31;5(10):1119-1126. doi: 10.1002/jgh3.12643. eCollection 2021 Oct. JGH Open. 2021. PMID: 34621996 Free PMC article. Review.
-
Long-term tolerability of topical diclofenac sodium 1% gel for osteoarthritis in seniors and patients with comorbidities.Clin Interv Aging. 2012;7:517-23. doi: 10.2147/CIA.S35416. Epub 2012 Nov 20. Clin Interv Aging. 2012. PMID: 23204844 Free PMC article. Clinical Trial.
-
Recent developments in crystal-induced inflammation pathogenesis and management.Curr Rheumatol Rep. 2007 Jun;9(3):243-50. doi: 10.1007/s11926-007-0039-5. Curr Rheumatol Rep. 2007. PMID: 17531179 Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

