Murine cytomegalovirus with a transposon insertional mutation at open reading frame m155 is deficient in growth and virulence in mice
- PMID: 15194765
- PMCID: PMC421665
- DOI: 10.1128/JVI.78.13.6891-6899.2004
Murine cytomegalovirus with a transposon insertional mutation at open reading frame m155 is deficient in growth and virulence in mice
Abstract
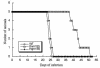
A pool of murine cytomegalovirus (MCMV) mutants was previously generated by using a Tn3-based transposon mutagenesis approach (X. Zhan, M. Lee, J. Xiao, and F. Liu, J. Virol. 74:7411-7421, 2000). In this study, one of the MCMV mutants, Rvm155, which contained the transposon insertion in open reading frame m155, was characterized in vitro for its replication in tissue culture and in vivo for its growth and virulence in immunodeficient SCID mice. Compared to the wild-type strain and a rescued virus that restored the m155 region, the mutant is significantly deficient in growth in many organs of the infected animals. At 21 days postinfection the titers of Rvm155 in the salivary glands, lungs, spleens, livers, and kidneys of the intraperitoneally infected SCID mice were lower than the titers of the wild-type virus and the rescued virus by 50-, 1,000-, 500-, 100-, and 500-fold, respectively. Moreover, the viral mutant was attenuated in killing the SCID mice, as none of the SCID mice that were intraperitoneally infected with Rvm155 died until 38 days postinfection while all the animals infected with the wild-type and rescued viruses died at 27 days postinfection. Our results provide the first direct evidence that a disruption of m155 expression leads to attenuation of viral virulence and growth in animals. Moreover, these results suggest that m155 is a viral determinant for optimal MCMV growth and virulence in vivo.
Figures


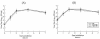
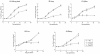

Similar articles
-
Murine cytomegalovirus open reading frame M27 plays an important role in growth and virulence in mice.J Virol. 2001 Feb;75(4):1697-707. doi: 10.1128/JVI.75.4.1697-1707.2001. J Virol. 2001. PMID: 11160668 Free PMC article.
-
In vitro and in vivo characterization of a murine cytomegalovirus with a mutation at open reading frame m166.J Virol. 2003 Mar;77(5):2882-91. doi: 10.1128/jvi.77.5.2882-2891.2003. J Virol. 2003. PMID: 12584312 Free PMC article.
-
Murine cytomegalovirus with a transposon insertional mutation at open reading frame M35 is defective in growth in vivo.J Virol. 2003 Jul;77(14):7746-55. doi: 10.1128/jvi.77.14.7746-7755.2003. J Virol. 2003. PMID: 12829814 Free PMC article.
-
Genetic analyses of gene function and pathogenesis of murine cytomegalovirus by transposon-mediated mutagenesis.J Clin Virol. 2002 Aug;25 Suppl 2:S111-22. doi: 10.1016/s1386-6532(02)00096-3. J Clin Virol. 2002. PMID: 12361762 Review.
-
Conditional gene expression systems to study herpesvirus biology in vivo.Med Microbiol Immunol. 2008 Jun;197(2):269-76. doi: 10.1007/s00430-008-0086-1. Epub 2008 Mar 7. Med Microbiol Immunol. 2008. PMID: 18324415 Review.
Cited by
-
Effective inhibition in animals of viral pathogenesis by a ribozyme derived from RNase P catalytic RNA.Proc Natl Acad Sci U S A. 2008 Aug 5;105(31):10919-24. doi: 10.1073/pnas.0804922105. Epub 2008 Jul 28. Proc Natl Acad Sci U S A. 2008. PMID: 18663226 Free PMC article.
-
Characterization of a highly glycosylated form of the human cytomegalovirus HLA class I homologue gpUL18.J Gen Virol. 2005 Nov;86(Pt 11):2999-3008. doi: 10.1099/vir.0.81126-0. J Gen Virol. 2005. PMID: 16227221 Free PMC article.
-
Selective down-regulation of the NKG2D ligand H60 by mouse cytomegalovirus m155 glycoprotein.J Virol. 2005 Mar;79(5):2920-30. doi: 10.1128/JVI.79.5.2920-2930.2005. J Virol. 2005. PMID: 15709011 Free PMC article.
-
Characterization of the murine cytomegalovirus m136 gene.Virus Genes. 2007 Apr;34(2):117-26. doi: 10.1007/s11262-006-0047-y. Epub 2006 Dec 2. Virus Genes. 2007. PMID: 17143724
-
Oral Vaccination with Attenuated Salmonella Expressing Viral M25 Protein Effectively Protects Mice Against Murine Cytomegalovirus Infection.Pathogens. 2025 Mar 25;14(4):314. doi: 10.3390/pathogens14040314. Pathogens. 2025. PMID: 40333046 Free PMC article.
References
-
- Arase, H., E. S. Mocarski, A. E. Campbell, A. B. Hill, and L. L. Lanier. 2002. Direct recognition of cytomegalovirus by activating and inhibitory NK cell receptors. Science 296:1323-1326. - PubMed
-
- Boname, J. M., and J. K. Chantler. 1992. Characterization of a strain of murine cytomegalovirus which fails to grow in the salivary glands of mice. J. Gen. Virol. 73:2021-2029. - PubMed
-
- Brune, W., C. Menard, U. Hobom, S. Odenbreit, M. Messerle, and U. H. Koszinowski. 1999. Rapid identification of essential and nonessential herpesvirus genes by direct transposon mutagenesis. Nat. Biotechnol. 17:360-364. - PubMed
-
- Chee, M. S., A. T. Bankier, S. Beck, R. Bohni, C. M. Brown, R. Cerny, T. Horsnell, C. A. Hutchison, T. Kouzarides, and J. A. Martignetti. 1990. Analysis of the protein-coding content of the sequence of human cytomegalovirus strain AD169. Curr. Top. Microbiol. Immunol. 154:125-169. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

