Cerebellar ataxia and Purkinje cell dysfunction caused by Ca2+-activated K+ channel deficiency
- PMID: 15194823
- PMCID: PMC439001
- DOI: 10.1073/pnas.0401702101
Cerebellar ataxia and Purkinje cell dysfunction caused by Ca2+-activated K+ channel deficiency
Abstract
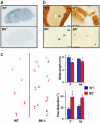
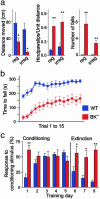
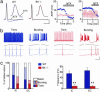
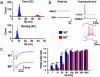
Malfunctions of potassium channels are increasingly implicated as causes of neurological disorders. However, the functional roles of the large-conductance voltage- and Ca(2+)-activated K(+) channel (BK channel), a unique calcium, and voltage-activated potassium channel type have remained elusive. Here we report that mice lacking BK channels (BK(-/-)) show cerebellar dysfunction in the form of abnormal conditioned eye-blink reflex, abnormal locomotion and pronounced deficiency in motor coordination, which are likely consequences of cerebellar learning deficiency. At the cellular level, the BK(-/-) mice showed a dramatic reduction in spontaneous activity of the BK(-/-) cerebellar Purkinje neurons, which generate the sole output of the cerebellar cortex and, in addition, enhanced short-term depression at the only output synapses of the cerebellar cortex, in the deep cerebellar nuclei. The impairing cellular effects caused by the lack of postsynaptic BK channels were found to be due to depolarization-induced inactivation of the action potential mechanism. These results identify previously unknown roles of potassium channels in mammalian cerebellar function and motor control. In addition, they provide a previously undescribed animal model of cerebellar ataxia.
Figures
Similar articles
-
Characterization of large conductance Ca2+-activated K+ channels in cerebellar Purkinje neurons.Eur J Neurosci. 2002 Oct;16(7):1214-22. doi: 10.1046/j.1460-9568.2002.02171.x. Eur J Neurosci. 2002. PMID: 12405981
-
Distinct contributions of small and large conductance Ca2+-activated K+ channels to rat Purkinje neuron function.J Physiol. 2003 Apr 1;548(Pt 1):53-69. doi: 10.1113/jphysiol.2002.027854. Epub 2003 Feb 7. J Physiol. 2003. PMID: 12576503 Free PMC article.
-
Discovery of Novel Activators of Large-Conductance Calcium-Activated Potassium Channels for the Treatment of Cerebellar Ataxia.Mol Pharmacol. 2022 Jul;102(1):438-449. doi: 10.1124/molpharm.121.000478. Epub 2022 Apr 30. Mol Pharmacol. 2022. PMID: 35489717 Free PMC article.
-
Large-conductance Ca2+- activated K+ channels:physiological role and pharmacology.Curr Med Chem. 2003 Apr;10(8):649-61. doi: 10.2174/0929867033457863. Curr Med Chem. 2003. PMID: 12678784 Review.
-
Calcium-activated potassium channels in adrenal chromaffin cells.Ion Channels. 1996;4:261-301. doi: 10.1007/978-1-4899-1775-1_7. Ion Channels. 1996. PMID: 8744211 Review.
Cited by
-
BK potassium channels facilitate high-frequency firing and cause early spike frequency adaptation in rat CA1 hippocampal pyramidal cells.J Physiol. 2007 May 1;580(Pt.3):859-82. doi: 10.1113/jphysiol.2006.126367. Epub 2007 Feb 15. J Physiol. 2007. PMID: 17303637 Free PMC article.
-
Stargazer--a mouse to seize!Epilepsy Curr. 2005 Sep-Oct;5(5):161-5. doi: 10.1111/j.1535-7511.2005.00051.x. Epilepsy Curr. 2005. PMID: 16175212 Free PMC article.
-
NaV1.1 channels and epilepsy.J Physiol. 2010 Jun 1;588(Pt 11):1849-59. doi: 10.1113/jphysiol.2010.187484. Epub 2010 Mar 1. J Physiol. 2010. PMID: 20194124 Free PMC article. Review.
-
Ca2+- and Voltage-Activated K+ (BK) Channels in the Nervous System: One Gene, a Myriad of Physiological Functions.Int J Mol Sci. 2023 Feb 8;24(4):3407. doi: 10.3390/ijms24043407. Int J Mol Sci. 2023. PMID: 36834817 Free PMC article. Review.
-
Maternal diabetes increases large conductance Ca2+-activated K+ outward currents that alter action potential properties but do not contribute to attenuated excitability of parasympathetic cardiac motoneurons in the nucleus ambiguus of neonatal mice.Am J Physiol Regul Integr Comp Physiol. 2011 May;300(5):R1070-8. doi: 10.1152/ajpregu.00470.2010. Epub 2011 Jan 19. Am J Physiol Regul Integr Comp Physiol. 2011. PMID: 21248308 Free PMC article.
References
-
- Hille, B. (2001) in Ion Channels of Excitable Membranes (Sinauer, Sunderland, MA), pp. 131-168.
-
- Browne, D. L., Gancher, S. T., Nutt, J. G., Brunt, E. R., Smith, E. A., Kramer, P. & Litt, M. (1994) Nat. Genet. 8, 136-140. - PubMed
-
- Herson, P. S. (2003) Nat. Neurosci. 6, 378-383. - PubMed
-
- Singh, N. A., Charlier, C., Stauffer, D., DuPont, B.R., Leach, R. J., Melis, R., Ronen, G. M., Bjerre, I., Quattlebaum, T., Murphy, J. V., et al. (1998) Nat. Genet. 18, 25-29. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

