Cellular immune responses induced in cattle by heterologous prime-boost vaccination using recombinant viruses and bacille Calmette-Guérin
- PMID: 15196215
- PMCID: PMC1782509
- DOI: 10.1111/j.1365-2567.2004.01903.x
Cellular immune responses induced in cattle by heterologous prime-boost vaccination using recombinant viruses and bacille Calmette-Guérin
Abstract
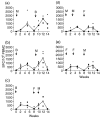
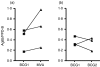
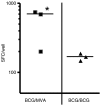
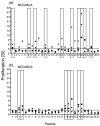
The development of novel vaccine strategies to replace or supplement bacille Calmette-Guérin (BCG) is urgently required. Here we study, in cattle, the use of heterologous prime-boost strategies based on vaccination with BCG and the mycobacterial mycolyl transferase Ag85A (Rv3804c) expressed either in recombinant modified vaccinia virus Ankara (MVA85A) or attenuated fowlpox strain FP9 (FP85A). Five different vaccination schedules were tested in the first experiment: MVA85A followed by BCG (group 1); BCG followed by MVA85A (group 2); BCG followed by FP85A and then MVA85A (group 3); MVA85A followed by MVA85A and then FP85A (group 4); and FP85A followed by FP85A and then MVA85A (group 5). Vaccine-induced levels of cellular immunity were assessed by determining interferon-gamma (IFN-gamma) responses in vitro. Prime-boost protocols, using recombinant MVA and BCG in combination (groups 1-3), resulted in significantly higher frequencies of Ag85-specific IFN-gamma-secreting cells than the two viral vectors used in combination (P=0.0055), or BCG used alone (groups 2 and 3, P=0.04). The T-cell repertoires of the calves in all five groups were significantly broader following heterologous booster immunizations than after the primary immunization. In a second experiment, the effects of BCG\MVA85A heterologous prime-boost vaccination were compared with BCG\BCG homologous revaccination. The results suggested a higher Ag85A-specific response with a wider T-cell repertoire in the MVA85A-boosted calves than in the BCG\BCG-vaccinated calves. In conclusion therefore, the present report demonstrates the effectiveness of heterologous prime-boost strategies based on recombinant MVA and BCG to induce strong cellular immune responses in cattle and prioritise such vaccination strategies for rapid assessment of protective efficacy in this natural target species of tuberculosis.
Figures

Similar articles
-
Alternate aerosol and systemic immunisation with a recombinant viral vector for tuberculosis, MVA85A: A phase I randomised controlled trial.PLoS Med. 2019 Apr 30;16(4):e1002790. doi: 10.1371/journal.pmed.1002790. eCollection 2019 Apr. PLoS Med. 2019. PMID: 31039172 Free PMC article. Clinical Trial.
-
Viral booster vaccines improve Mycobacterium bovis BCG-induced protection against bovine tuberculosis.Infect Immun. 2009 Aug;77(8):3364-73. doi: 10.1128/IAI.00287-09. Epub 2009 Jun 1. Infect Immun. 2009. PMID: 19487476 Free PMC article.
-
Heterologous boosting with recombinant VSV-846 in BCG-primed mice confers improved protection against Mycobacterium infection.Hum Vaccin Immunother. 2017 Apr 3;13(4):816-822. doi: 10.1080/21645515.2016.1261229. Epub 2016 Dec 14. Hum Vaccin Immunother. 2017. PMID: 27960596 Free PMC article.
-
Synergistic DNA-MVA prime-boost vaccination regimes for malaria and tuberculosis.Vaccine. 2006 May 22;24(21):4554-61. doi: 10.1016/j.vaccine.2005.08.048. Epub 2005 Aug 24. Vaccine. 2006. PMID: 16150517 Review.
-
Heterologous boost vaccines for bacillus Calmette-Guérin prime immunization against tuberculosis.Expert Rev Vaccines. 2007 Aug;6(4):539-46. doi: 10.1586/14760584.6.4.539. Expert Rev Vaccines. 2007. PMID: 17669008 Review.
Cited by
-
The evolution of poxvirus vaccines.Viruses. 2015 Apr 7;7(4):1726-803. doi: 10.3390/v7041726. Viruses. 2015. PMID: 25853483 Free PMC article. Review.
-
Immune responses induced in cattle by vaccination with a recombinant adenovirus expressing Mycobacterial antigen 85A and Mycobacterium bovis BCG.Infect Immun. 2006 Feb;74(2):1416-8. doi: 10.1128/IAI.74.2.1416-1418.2006. Infect Immun. 2006. PMID: 16428796 Free PMC article.
-
Establishment of an aerosol challenge model of tuberculosis in rhesus macaques and an evaluation of endpoints for vaccine testing.Clin Vaccine Immunol. 2010 Aug;17(8):1170-82. doi: 10.1128/CVI.00079-10. Epub 2010 Jun 9. Clin Vaccine Immunol. 2010. PMID: 20534795 Free PMC article.
-
Flow cytometric detection of gamma interferon can effectively discriminate Mycobacterium bovis BCG-vaccinated cattle from M. bovis-infected cattle.Clin Vaccine Immunol. 2006 Dec;13(12):1343-8. doi: 10.1128/CVI.00291-06. Epub 2006 Sep 27. Clin Vaccine Immunol. 2006. PMID: 17005929 Free PMC article.
-
Immunogenicity and protective efficacy against murine tuberculosis of a prime-boost regimen with BCG and a DNA vaccine expressing ESAT-6 and Ag85A fusion protein.Clin Dev Immunol. 2011;2011:617892. doi: 10.1155/2011/617892. Epub 2011 Feb 27. Clin Dev Immunol. 2011. PMID: 21461375 Free PMC article.
References
-
- Steele JH. Regional and Country Status Report. In: Thoen CO, Steele JH, editors. Mycobacterium bovis Infection in Animals and Humans. Ames: Iowa State University Press; 1995. pp. 169–72.
-
- Krebs JR. Bovine Tuberculosis in Cattle and Badgers. London, UK: Ministry of Agriculture, Fisheries and Food Publications; 1997.
-
- Hewinson RG, Vordermeier HM, Buddle BM. Use of the bovine model of tuberculosis for the development of improved vaccines and diagnostics. Tuberculosis. 2003;83:119–30. - PubMed
-
- Fine PE. The BCG story: lessons from the past and implications for the future. Rev Infect Dis. 1989;11(Suppl. 2):S353–9. - PubMed
-
- Francis J. Bovine Tuberculosis. London: Staples Press; 1947.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
