Targeted inactivation of testicular nuclear orphan receptor 4 delays and disrupts late meiotic prophase and subsequent meiotic divisions of spermatogenesis
- PMID: 15199144
- PMCID: PMC480911
- DOI: 10.1128/MCB.24.13.5887-5899.2004
Targeted inactivation of testicular nuclear orphan receptor 4 delays and disrupts late meiotic prophase and subsequent meiotic divisions of spermatogenesis
Abstract
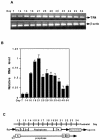
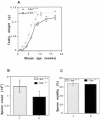
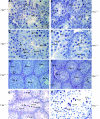
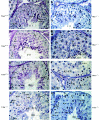
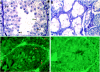
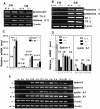
Testicular orphan nuclear receptor 4 (TR4) is specifically and stage-dependently expressed in late-stage pachytene spermatocytes and round spermatids. In the developing mouse testis, the highest expression of TR4 can be detected at postnatal days 16 to 21 when the first wave of spermatogenesis progresses to late meiotic prophase. Using a knockout strategy to delete TR4 in mice, we found that sperm production in TR4(-/-) mice is reduced. The comparison of testes from developing TR4(+/+) and TR4(-/-) mice shows that spermatogenesis in TR4(-/-) mice is delayed. Analysis of the first wave of spermatogenesis shows that the delay can be due to delay and disruption of spermatogenesis at the end of late meiotic prophase and subsequent meiotic divisions. Seminiferous tubule staging shows that stages X to XII, where late meiotic prophase and meiotic divisions take place, are delayed and disrupted in TR4(-/-) mice. Histological examination of testis sections from TR4(-/-) mice shows degenerated primary spermatocytes and some necrotic tubules. Testis-specific gene analyses show that the expression of sperm 1 and cyclin A1, which are genes expressed at the end of meiotic prophase, was delayed and decreased in TR4(-/-) mouse testes. Taken together, results from TR4(+/+) and TR4(-/-) mice indicate that TR4 is essential for normal spermatogenesis in mice.
Figures

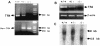




Similar articles
-
Expression of orphan receptors TR2, TR3, TR4, and p53 in heat-treated testis of cynomolgus monkeys (Macaca fascicularis).J Androl. 2006 May-Jun;27(3):405-13. doi: 10.2164/jandrol.05165. Epub 2006 Feb 1. J Androl. 2006. PMID: 16452526
-
Spermatogenesis and testis development are normal in mice lacking testicular orphan nuclear receptor 2.Mol Cell Biol. 2002 Jul;22(13):4661-6. doi: 10.1128/MCB.22.13.4661-4666.2002. Mol Cell Biol. 2002. PMID: 12052874 Free PMC article.
-
Inactivation of insulin-like factor 6 disrupts the progression of spermatogenesis at late meiotic prophase.Endocrinology. 2009 Sep;150(9):4348-57. doi: 10.1210/en.2009-0201. Epub 2009 Jun 11. Endocrinology. 2009. PMID: 19520787
-
Surfing the wave, cycle, life history, and genes/proteins expressed by testicular germ cells. Part 1: background to spermatogenesis, spermatogonia, and spermatocytes.Microsc Res Tech. 2010 Apr;73(4):241-78. doi: 10.1002/jemt.20783. Microsc Res Tech. 2010. PMID: 19941293 Review.
-
MEIOSIN directs initiation of meiosis and subsequent meiotic prophase program during spermatogenesis.Genes Genet Syst. 2022 Jun 4;97(1):27-39. doi: 10.1266/ggs.21-00054. Epub 2021 Dec 25. Genes Genet Syst. 2022. PMID: 34955498 Review.
Cited by
-
Roles of Nuclear Orphan Receptors TR2 and TR4 during Hematopoiesis.Genes (Basel). 2024 Apr 27;15(5):563. doi: 10.3390/genes15050563. Genes (Basel). 2024. PMID: 38790192 Free PMC article. Review.
-
Fidgetin-like1 is a strong candidate for a dynamic impairment of male meiosis leading to reduced testis weight in mice.PLoS One. 2011;6(11):e27582. doi: 10.1371/journal.pone.0027582. Epub 2011 Nov 16. PLoS One. 2011. PMID: 22110678 Free PMC article.
-
Disrupted Gene Networks in Subfertile Hybrid House Mice.Mol Biol Evol. 2020 Jun 1;37(6):1547-1562. doi: 10.1093/molbev/msaa002. Mol Biol Evol. 2020. PMID: 32076722 Free PMC article.
-
Adult functions for the Drosophila DHR78 nuclear receptor.Dev Dyn. 2018 Feb;247(2):315-322. doi: 10.1002/dvdy.24608. Epub 2017 Dec 12. Dev Dyn. 2018. PMID: 29171103 Free PMC article.
-
The nuclear receptors of Biomphalaria glabrata and Lottia gigantea: implications for developing new model organisms.PLoS One. 2015 Apr 7;10(4):e0121259. doi: 10.1371/journal.pone.0121259. eCollection 2015. PLoS One. 2015. PMID: 25849443 Free PMC article.
References
-
- Dix, D. J., J. W. Allen, B. W. Collin, P. Poorman-Allen, C. Mori, D. R. Blizard, P. R. Brown, E. H. Goulding, B. D. Strong, and E. M. Eddy. 1997. HSP70-2 is required for desynapsis of synaponemal complexes during meiotic prophase in juvenile and adult mouse spermatocytes. Development 124:4595-4605. - PubMed
-
- Drabent, B., C. Bode, B. Bramlage, and D. Doenecke. 1996. Expression of the mouse testicular histone gene H1t during spermatogenesis. Histochem. Cell Biol. 106:247-251. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
