Induction of dominant transplantation tolerance by an altered peptide ligand of the male antigen Dby
- PMID: 15199410
- PMCID: PMC420506
- DOI: 10.1172/JCI20569
Induction of dominant transplantation tolerance by an altered peptide ligand of the male antigen Dby
Abstract
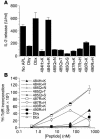
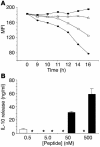
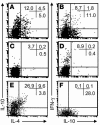
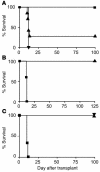
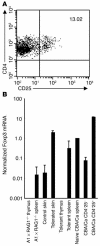
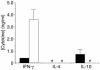
T cell reactivity to minor histocompatibility (mH) antigens is responsible for rejection of HLA-matched allografts, limiting the effectiveness of transplantation for the treatment of end-stage organ failure. The deadbox gene Dby is located on the Y chromosome and encodes an mH antigen that prompts rejection of male tissues by female mice. Establishing a network of regulatory T (T(reg)) cells that is capable of coercing naive cells to adopt a tolerant phenotype offers an attractive strategy for immune intervention in such deleterious immune responses. While various approaches have successfully induced a dominant form of transplantation tolerance, they share the propensity to provoke chronic, incomplete activation of T cells. By identifying the T cell receptor (TCR) contact sites of the dominant epitope of the Dby gene product, we have designed an altered peptide ligand (APL) that delivers incomplete signals to naive T cells from A1 infinity RAG1(-/-) mice that are transgenic for a complementary TCR. Administration of this APL to female transgenic mice polarizes T cells toward a regulatory phenotype, securing a form of dominant tolerance to male skin grafts that is capable of resisting rejection by naive lymphocytes. Our results demonstrate that incomplete signaling through the TCR may establish a network of T(reg) cells that may be harnessed in the service of transplantation tolerance.
Figures
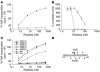
Comment in
-
Is transplantation tolerable?J Clin Invest. 2004 Jun;113(12):1681-3. doi: 10.1172/JCI22153. J Clin Invest. 2004. PMID: 15199402 Free PMC article.
References
-
- Goulmy E. Human minor histocompatibility antigens: new concepts for marrow transplantation and adoptive immunotherapy. Immunol. Rev. 1997;157:125–140. - PubMed
-
- Sakaguchi S, et al. Immunologic tolerance maintained by CD25+CD4+ regulatory T cells: their common role in controlling autoimmunity, tumor immunity, and transplantation tolerance. Immunol. Rev. 2001;182:18–32. - PubMed
-
- Graça L, et al. Both CD4+25+ and CD4+25- regulatory T cells mediate dominant transplantation tolerance. J. Immunol. 2002;168:5558–5565. - PubMed
-
- Waldmann H. Reprogramming the immune system. Immunol. Rev. 2002;185:227–235. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

