Kaposi's sarcoma-associated herpesvirus induction of chromosome instability in primary human endothelial cells
- PMID: 15205312
- PMCID: PMC5257260
- DOI: 10.1158/0008-5472.CAN-04-0657
Kaposi's sarcoma-associated herpesvirus induction of chromosome instability in primary human endothelial cells
Abstract
Chromosome instability contributes to the multistep oncogenesis of cancer cells. Kaposi's sarcoma (KS), an angiogenic vascular spindle cancer of endothelial cells, displays stage advancement with lesions at early stage being hyperproliferative, whereas lesions at late stage are clonal or multiclonal and can exhibit a neoplastic nature and chromosome instability. Although infection with KS-associated herpesvirus (KSHV) has been associated with the initiation and promotion of KS, the mechanism of KS neoplastic transformation remains unclear. We show that KSHV infection of primary human umbilical vein endothelial cells induces abnormal mitotic spindles and centrosome duplication. As a result, KSHV-infected cells manifest chromosome instability, including chromosomal misalignments and laggings, mitotic bridges, and formation of micronuclei and multinucleation. Our results indicate that KSHV infection could predispose cells to malignant transformation through induction of genomic instability and contributes to the development of KS.
Figures
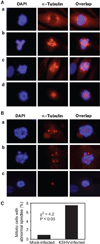
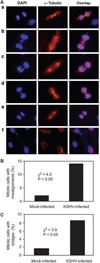

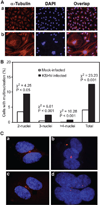
Similar articles
-
Kaposi's sarcoma-associated herpesvirus induces sustained levels of vascular endothelial growth factors A and C early during in vitro infection of human microvascular dermal endothelial cells: biological implications.J Virol. 2008 Feb;82(4):1759-76. doi: 10.1128/JVI.00873-07. Epub 2007 Dec 5. J Virol. 2008. PMID: 18057235 Free PMC article.
-
Spindle cell conversion by Kaposi's sarcoma-associated herpesvirus: formation of colonies and plaques with mixed lytic and latent gene expression in infected primary dermal microvascular endothelial cell cultures.J Virol. 2001 Jun;75(12):5614-26. doi: 10.1128/JVI.75.12.5614-5626.2001. J Virol. 2001. PMID: 11356969 Free PMC article.
-
Does dysregulated expression of a deregulated viral GPCR trigger Kaposi's sarcomagenesis?FASEB J. 2004 Mar;18(3):422-7. doi: 10.1096/fj.03-1035hyp. FASEB J. 2004. PMID: 15003988
-
The Kaposi's sarcoma-associated herpesvirus (human herpesvirus-8) in Kaposi's sarcoma, malignant lymphoma, and other diseases.Ann Oncol. 1997;8 Suppl 2:123-9. Ann Oncol. 1997. PMID: 9209655 Review.
-
Kaposi's sarcoma: a model of both malignancy and chronic inflammation.Panminerva Med. 2007 Sep;49(3):119-38. Panminerva Med. 2007. PMID: 17912148 Review.
Cited by
-
Kaposi's Sarcoma-Associated Herpesvirus Genome Replication, Partitioning, and Maintenance in Latency.Front Microbiol. 2012 Jan 24;3:7. doi: 10.3389/fmicb.2012.00007. eCollection 2012. Front Microbiol. 2012. PMID: 22291692 Free PMC article.
-
HTLV-1 Tax: centrosome amplification and cancer.Retrovirology. 2006 Aug 9;3:50. doi: 10.1186/1742-4690-3-50. Retrovirology. 2006. PMID: 16899128 Free PMC article.
-
Koilocytes are enriched for alkaline-labile sites.Eur J Histochem. 2010;54(4):e32. doi: 10.4081/ejh.2010.e32. Eur J Histochem. 2010. PMID: 21337807 Free PMC article.
-
R Loops and Links to Human Disease.J Mol Biol. 2017 Oct 27;429(21):3168-3180. doi: 10.1016/j.jmb.2016.08.031. Epub 2016 Sep 4. J Mol Biol. 2017. PMID: 27600412 Free PMC article. Review.
-
Latency-associated nuclear antigen of Kaposi's sarcoma-associated herpesvirus recruits uracil DNA glycosylase 2 at the terminal repeats and is important for latent persistence of the virus.J Virol. 2006 Nov;80(22):11178-90. doi: 10.1128/JVI.01334-06. Epub 2006 Aug 23. J Virol. 2006. PMID: 16928741 Free PMC article.
References
-
- Gisselsson D. Chromosome instability in cancer: how, when, and why. Adv Cancer Res. 2003:1–29. - PubMed
-
- Lavia P, Mileo AM, Giordano A, Paggi MG. Emerging roles of DNA tumor viruses in cell proliferation: new insights into genomic instability. Oncogene. 2003;22:6508–6516. - PubMed
-
- Fortunato EA, Spector DH. Viral induction of site-specific chromosome damage. Rev Med Virol. 2003;13:21–37. - PubMed
-
- Steffensen DM, Szabo P, McDougall JK. Adenovirus 12 uncoiler region of human chromosome 1 in relation to the 5s rRNA genes. Exp Cell Res. 1976;100:436–439. - PubMed
-
- Luleci G, Sakizli M, Gunalp A. Selective chromosome damage caused by human cytomegalovirus. Acta Virol. 1980;24:341–345. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

