Candida parapsilosis characterization in an outbreak setting
- PMID: 15207060
- PMCID: PMC3323144
- DOI: 10.3201/eid1006.030873
Candida parapsilosis characterization in an outbreak setting
Abstract
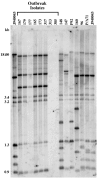
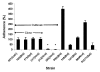
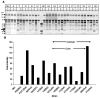
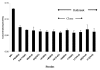
Candida parapsilosis is an important non-albicans species which infects hospitalized patients. No studies have correlated outbreak infections of C. parapsilosis with multiple virulence factors. We used DNA fingerprinting to determine genetic variability among isolates from a C. parapsilosis outbreak and from our clinical database. We compared phenotypic markers of pathogenesis, including adherence, biofilm formation, and protein secretion (secretory aspartic protease [SAP] and phospholipase). Adherence was measured as colony counts on silicone elastomer disks immersed in agar. Biofilms formed on disks were quantified by dry weight. SAP expression was measured by hydrolysis of bovine albumin; a colorimetric assay was used to quantitate phospholipase. DNA fingerprinting indicated that the outbreak isolates were clonal and genetically distinct from our database. Biofilm expression by the outbreak clone was greater than that of sporadic isolates (p < or =0.0005). Adherence and protein secretion did not correlate with strain pathogenicity. These results suggest that biofilm production plays a role in C. parapsilosis outbreaks.
Figures


Similar articles
-
Acid proteinase, phospholipase, and biofilm production of Candida species isolated from blood cultures.Mycopathologia. 2007 Dec;164(6):265-9. doi: 10.1007/s11046-007-9053-4. Epub 2007 Sep 15. Mycopathologia. 2007. PMID: 17874282
-
Molecular epidemiology and antifungal susceptibility of Candida parapsilosis sensu stricto, Candida orthopsilosis, and Candida metapsilosis in Taiwan.Diagn Microbiol Infect Dis. 2010 Nov;68(3):284-92. doi: 10.1016/j.diagmicrobio.2010.07.004. Epub 2010 Sep 20. Diagn Microbiol Infect Dis. 2010. PMID: 20851551
-
Simple, Low-Cost Detection of Candida parapsilosis Complex Isolates and Molecular Fingerprinting of Candida orthopsilosis Strains in Kuwait by ITS Region Sequencing and Amplified Fragment Length Polymorphism Analysis.PLoS One. 2015 Nov 18;10(11):e0142880. doi: 10.1371/journal.pone.0142880. eCollection 2015. PLoS One. 2015. PMID: 26580965 Free PMC article.
-
Adherence and biofilm formation of non-Candida albicans Candida species.Trends Microbiol. 2011 May;19(5):241-7. doi: 10.1016/j.tim.2011.02.003. Epub 2011 Mar 15. Trends Microbiol. 2011. PMID: 21411325 Review.
-
Candida survival strategies.Adv Appl Microbiol. 2015;91:139-235. doi: 10.1016/bs.aambs.2014.12.002. Epub 2015 Feb 24. Adv Appl Microbiol. 2015. PMID: 25911234 Review.
Cited by
-
Epidemiology, risk factors, and prognosis of Candida parapsilosis bloodstream infections: case-control population-based surveillance study of patients in Barcelona, Spain, from 2002 to 2003.J Clin Microbiol. 2006 May;44(5):1681-5. doi: 10.1128/JCM.44.5.1681-1685.2006. J Clin Microbiol. 2006. PMID: 16672393 Free PMC article.
-
Anidulafungin versus caspofungin in a mouse model of candidiasis caused by anidulafungin-susceptible Candida parapsilosis isolates with different degrees of caspofungin susceptibility.Antimicrob Agents Chemother. 2014;58(1):229-36. doi: 10.1128/AAC.01025-13. Epub 2013 Oct 21. Antimicrob Agents Chemother. 2014. PMID: 24145540 Free PMC article.
-
Correlation between biofilm formation and the hypoxic response in Candida parapsilosis.Eukaryot Cell. 2009 Apr;8(4):550-9. doi: 10.1128/EC.00350-08. Epub 2009 Jan 16. Eukaryot Cell. 2009. PMID: 19151323 Free PMC article.
-
Species-specific and drug-specific differences in susceptibility of Candida biofilms to echinocandins: characterization of less common bloodstream isolates.Antimicrob Agents Chemother. 2013 Jun;57(6):2562-70. doi: 10.1128/AAC.02541-12. Epub 2013 Mar 25. Antimicrob Agents Chemother. 2013. PMID: 23529739 Free PMC article.
-
A single-centre 10-year experience with Candida bloodstream infections.Can J Infect Dis Med Microbiol. 2009 Summer;20(2):45-50. doi: 10.1155/2009/731070. Can J Infect Dis Med Microbiol. 2009. PMID: 20514159 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
