Efficient assembly of photosystem II in Chlamydomonas reinhardtii requires Alb3.1p, a homolog of Arabidopsis ALBINO3
- PMID: 15208384
- PMCID: PMC514161
- DOI: 10.1105/tpc.023226
Efficient assembly of photosystem II in Chlamydomonas reinhardtii requires Alb3.1p, a homolog of Arabidopsis ALBINO3
Abstract
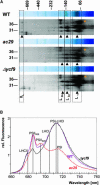
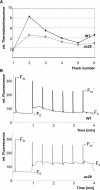
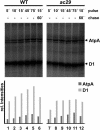
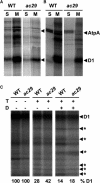
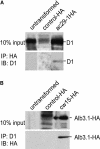
Alb3 homologs Oxa1 and YidC have been shown to be required for the integration of newly synthesized proteins into membranes. Here, we show that although Alb3.1p is not required for integration of the plastid-encoded photosystem II core subunit D1 into the thylakoid membrane of Chlamydomonas reinhardtii, the insertion of D1 into functional photosystem II complexes is retarded in the Alb3.1 deletion mutant ac29. Alb3.1p is associated with D1 upon its insertion into the membrane, indicating that Alb3.1p is essential for the efficient assembly of photosystem II. Furthermore, levels of nucleus-encoded light-harvesting proteins are vastly reduced in ac29; however, the remaining antenna systems are still connected to photosystem II reaction centers. Thus, Alb3.1p has a dual function and is required for the accumulation of both nucleus- and plastid-encoded protein subunits in photosynthetic complexes of C. reinhardtii.
Figures


Similar articles
-
One of two alb3 proteins is essential for the assembly of the photosystems and for cell survival in Chlamydomonas.Plant Cell. 2006 Jun;18(6):1454-66. doi: 10.1105/tpc.105.038695. Epub 2006 May 5. Plant Cell. 2006. PMID: 16679460 Free PMC article.
-
Assembly Apparatus of Light-Harvesting Complexes: Identification of Alb3.1-cpSRP-LHCP Complexes in the Green Alga Chlamydomonas reinhardtii.Plant Cell Physiol. 2022 Jan 25;63(1):70-81. doi: 10.1093/pcp/pcab146. Plant Cell Physiol. 2022. PMID: 34592750
-
Loss of Albino3 leads to the specific depletion of the light-harvesting system.Plant Cell. 2002 Sep;14(9):2303-14. doi: 10.1105/tpc.003442. Plant Cell. 2002. PMID: 12215522 Free PMC article.
-
Environmentally modulated phosphorylation and dynamics of proteins in photosynthetic membranes.Biochim Biophys Acta. 2007 Jun;1767(6):449-57. doi: 10.1016/j.bbabio.2006.11.007. Epub 2006 Nov 15. Biochim Biophys Acta. 2007. PMID: 17184728 Review.
-
Possible role for molecular chaperones in assembly and repair of photosystem II.Biochem Soc Trans. 2001 Aug;29(Pt 4):413-8. doi: 10.1042/bst0290413. Biochem Soc Trans. 2001. PMID: 11497999 Review.
Cited by
-
Loss of the SPHF homologue Slr1768 leads to a catastrophic failure in the maintenance of thylakoid membranes in Synechocystis sp. PCC 6803.PLoS One. 2011;6(5):e19625. doi: 10.1371/journal.pone.0019625. Epub 2011 May 23. PLoS One. 2011. PMID: 21625427 Free PMC article.
-
Regulatory factors for the assembly of thylakoid membrane protein complexes.Philos Trans R Soc Lond B Biol Sci. 2012 Dec 19;367(1608):3420-9. doi: 10.1098/rstb.2012.0065. Philos Trans R Soc Lond B Biol Sci. 2012. PMID: 23148269 Free PMC article. Review.
-
Complexome profiling on the Chlamydomonas lpa2 mutant reveals insights into PSII biogenesis and new PSII associated proteins.J Exp Bot. 2022 Jan 5;73(1):245-262. doi: 10.1093/jxb/erab390. J Exp Bot. 2022. PMID: 34436580 Free PMC article.
-
One of two alb3 proteins is essential for the assembly of the photosystems and for cell survival in Chlamydomonas.Plant Cell. 2006 Jun;18(6):1454-66. doi: 10.1105/tpc.105.038695. Epub 2006 May 5. Plant Cell. 2006. PMID: 16679460 Free PMC article.
-
RNA-seq analysis of the effect of kanamycin and the ABC transporter AtWBC19 on Arabidopsis thaliana seedlings reveals changes in metal content.PLoS One. 2014 Oct 13;9(10):e109310. doi: 10.1371/journal.pone.0109310. eCollection 2014. PLoS One. 2014. PMID: 25310285 Free PMC article.
References
-
- Alfonso, M., Montoya, G., Cases, R., Rodriguez, R., and Picorel, R. (1994). Core antenna complexes, CP43 and CP47, of higher plant photosystem II. Spectral properties, pigment stoichiometry, and amino acid composition. Biochemistry 33, 10494–10500. - PubMed
-
- Chen, M., Xie, K., Jiang, F., Yi, L., and Dalbey, R.E. (2002). YidC, a newly defined evolutionarily conserved protein, mediates membrane protein assembly in bacteria. Biol. Chem. 383, 1565–1572. - PubMed
-
- Choquet, Y., and Wollman, F.A. (2002). Translational regulations as specific traits of chloroplast gene expression. FEBS Lett. 529, 39–42. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

