PrP(C) association with lipid rafts in the early secretory pathway stabilizes its cellular conformation
- PMID: 15229281
- PMCID: PMC515338
- DOI: 10.1091/mbc.e03-05-0271
PrP(C) association with lipid rafts in the early secretory pathway stabilizes its cellular conformation
Abstract
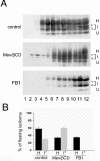
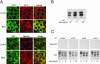
The pathological conversion of cellular prion protein (PrP(C)) into the scrapie prion protein (PrP(Sc)) isoform appears to have a central role in the pathogenesis of transmissible spongiform encephalopathies. However, the identity of the intracellular compartment where this conversion occurs is unknown. Several lines of evidence indicate that detergent-resistant membrane domains (DRMs or rafts) could be involved in this process. We have characterized the association of PrP(C) to rafts during its biosynthesis. We found that PrP(C) associates with rafts already as an immature precursor in the endoplasmic reticulum. Interestingly, compared with the mature protein, the immature diglycosylated form has a different susceptibility to cholesterol depletion vs. sphingolipid depletion, suggesting that the two forms associate with different lipid domains. We also found that cholesterol depletion, which affects raft-association of the immature protein, slows down protein maturation and leads to protein misfolding. On the contrary, sphingolipid depletion does not have any effect on the kinetics of protein maturation or on the conformation of the protein. These data indicate that the early association of PrP(C) with cholesterol-enriched rafts facilitates its correct folding and reinforce the hypothesis that cholesterol and sphingolipids have different roles in PrP metabolism.
Figures






Similar articles
-
Detergent-resistant membrane domains but not the proteasome are involved in the misfolding of a PrP mutant retained in the endoplasmic reticulum.J Cell Sci. 2006 Feb 1;119(Pt 3):433-42. doi: 10.1242/jcs.02768. J Cell Sci. 2006. PMID: 16443748
-
PrPC is sorted to the basolateral membrane of epithelial cells independently of its association with rafts.Traffic. 2002 Nov;3(11):810-21. doi: 10.1034/j.1600-0854.2002.31106.x. Traffic. 2002. PMID: 12383347
-
The prion protein and lipid rafts.Mol Membr Biol. 2006 Jan-Feb;23(1):89-99. doi: 10.1080/09687860500449994. Mol Membr Biol. 2006. PMID: 16611584 Review.
-
Glypican-1 mediates both prion protein lipid raft association and disease isoform formation.PLoS Pathog. 2009 Nov;5(11):e1000666. doi: 10.1371/journal.ppat.1000666. Epub 2009 Nov 20. PLoS Pathog. 2009. PMID: 19936054 Free PMC article.
-
The role of rafts in the fibrillization and aggregation of prions.Chem Phys Lipids. 2006 Jun;141(1-2):66-71. doi: 10.1016/j.chemphyslip.2006.02.022. Epub 2006 Mar 27. Chem Phys Lipids. 2006. PMID: 16647049 Review.
Cited by
-
Lipid rafts and clathrin cooperate in the internalization of PrP in epithelial FRT cells.PLoS One. 2009 Jun 8;4(6):e5829. doi: 10.1371/journal.pone.0005829. PLoS One. 2009. PMID: 19503793 Free PMC article.
-
One Raft to Guide Them All, and in Axon Regeneration Inhibit Them.Int J Mol Sci. 2021 May 8;22(9):5009. doi: 10.3390/ijms22095009. Int J Mol Sci. 2021. PMID: 34066896 Free PMC article. Review.
-
Membrane Domain Localization and Interaction of the Prion-Family Proteins, Prion and Shadoo with Calnexin.Membranes (Basel). 2021 Dec 13;11(12):978. doi: 10.3390/membranes11120978. Membranes (Basel). 2021. PMID: 34940479 Free PMC article.
-
The Multifaceted Functions of Prion Protein (PrPC) in Cancer.Cancers (Basel). 2023 Oct 13;15(20):4982. doi: 10.3390/cancers15204982. Cancers (Basel). 2023. PMID: 37894349 Free PMC article. Review.
-
Rab14 regulates apical targeting in polarized epithelial cells.Traffic. 2008 Jul;9(7):1218-31. doi: 10.1111/j.1600-0854.2008.00752.x. Epub 2008 Apr 21. Traffic. 2008. PMID: 18429929 Free PMC article.
References
-
- Bartlett, S.E., Reynolds, A.J., Weible, M., 2nd, Noakes, P.G., and Hendry, I.A. (2001). Transport of endosomal early antigen 1 in the rat sciatic nerve and location in cultured neurons. Neuroreport 12, 281-284. - PubMed
-
- Beranger, F., Mange, A., Goud, B., and Lehmann, S. (2002). Stimulation of PrP(C) retrograde transport toward the endoplasmic reticulum increases accumulation of PrP(Sc) in prion-infected cells. J. Biol. Chem. 277, 38972-38977. - PubMed
-
- Bogdanov, M., and Dowhan, W. (1999). Lipid-assisted protein folding. J. Biol. Chem. 274, 36827-36830. - PubMed
-
- Borchelt, D.R., Taraboulos, A., and Prusiner, S.B. (1992). Evidence for synthesis of scrapie prion proteins in the endocytic pathway. J. Biol. Chem. 267, 16188-16199. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

