Expression and functional activity of PPARgamma in pancreatic beta cells
- PMID: 15237101
- PMCID: PMC1575182
- DOI: 10.1038/sj.bjp.0705844
Expression and functional activity of PPARgamma in pancreatic beta cells
Abstract
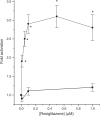
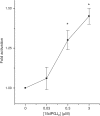
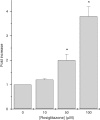
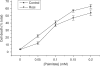
Rosiglitazone is an agonist of peroxisome proliferator activated receptor-gamma (PPARgamma) and ameliorates insulin resistance in type II diabetes. In addition, it may also promote increased pancreatic beta-cell viability, although it is not known whether this effect is mediated by a direct action on the beta cell. We have investigated this possibility. Semiquantitative real-time reverse transcription-polymerase chain reaction analysis (Taqman) revealed that freshly isolated rat islets and the clonal beta-cell line, BRIN-BD11, express PPARgamma, as well as PPARalpha and PPARdelta. The levels of expression of PPARgamma were estimated by reference to adipose tissue and were found to represent approximately 60% (islets) and 30% (BRIN-BD11) of that found in freshly isolated visceral adipose tissue. Western blotting confirmed the presence of immunoreactive PPARgamma in rat (and human) islets and in BRIN-BD11 cells. Transfection of BRIN-BD11 cells with a PPARgamma-sensitive luciferase reporter construct was used to evaluate the functional competence of the endogenous PPARgamma. Luciferase activity was modestly increased by the putative endogenous ligand, 15-deoxy-Delta12,14 prostaglandin J2 (15dPGJ2). Rosiglitazone also caused activation of the luciferase reporter construct but this effect required concentrations of the drug (50-100 microm) that are beyond the expected therapeutic range. This suggests that PPARgamma is relatively insensitive to activation by rosiglitazone in BRIN-BD11 cells. Exposure of BRIN-BD11 cells to the lipotoxic effector, palmitate, caused a marked loss of viability. This was attenuated by treatment of the cells with either actinomycin D or cycloheximide suggesting that a pathway of programmed cell death was involved. Rosiglitazone failed to protect BRIN-BD11 cells from the toxic actions of palmitate at concentrations up to 50 microm. Similar results were obtained with a range of other PPARgamma agonists. Taken together, the present data suggest that, at least under in vitro conditions, thiazolidinediones do not exert direct protective effects against fatty acid-mediated cytotoxicity in pancreatic beta cells.
Copyright 2004 Nature Publishing Group
Figures



Similar articles
-
A novel selective peroxisome proliferator-activator receptor-gamma modulator-SPPARgammaM5 improves insulin sensitivity with diminished adverse cardiovascular effects.Eur J Pharmacol. 2008 Apr 14;584(1):192-201. doi: 10.1016/j.ejphar.2007.12.036. Epub 2008 Feb 14. Eur J Pharmacol. 2008. PMID: 18346728
-
Rosiglitazone induces interleukin-1 receptor antagonist in interleukin-1beta-stimulated rat synovial fibroblasts via a peroxisome proliferator-activated receptor beta/delta-dependent mechanism.Arthritis Rheum. 2005 Mar;52(3):759-69. doi: 10.1002/art.20868. Arthritis Rheum. 2005. PMID: 15751073
-
Rosiglitazone prevents the impairment of human islet function induced by fatty acids: evidence for a role of PPARgamma2 in the modulation of insulin secretion.Am J Physiol Endocrinol Metab. 2004 Apr;286(4):E560-7. doi: 10.1152/ajpendo.00561.2002. Epub 2003 Nov 18. Am J Physiol Endocrinol Metab. 2004. PMID: 14625208
-
Peroxisome proliferator-activated receptors in vascular biology-molecular mechanisms and clinical implications.Vascul Pharmacol. 2006 Jul;45(1):19-28. doi: 10.1016/j.vph.2005.11.014. Epub 2006 Jun 16. Vascul Pharmacol. 2006. PMID: 16782410 Review.
-
Engineering cultured insulin-secreting pancreatic B-cell lines.J Mol Med (Berl). 1999 Jan;77(1):235-43. doi: 10.1007/s001090050344. J Mol Med (Berl). 1999. PMID: 9930971 Review.
Cited by
-
The role of peroxisome proliferator-activated receptor γ in pancreatic β cell function and survival: therapeutic implications for the treatment of type 2 diabetes mellitus.Diabetes Obes Metab. 2010 Dec;12(12):1036-47. doi: 10.1111/j.1463-1326.2010.01299.x. Diabetes Obes Metab. 2010. PMID: 20977574 Free PMC article.
-
Is the Mouse a Good Model of Human PPARγ-Related Metabolic Diseases?Int J Mol Sci. 2016 Jul 30;17(8):1236. doi: 10.3390/ijms17081236. Int J Mol Sci. 2016. PMID: 27483259 Free PMC article. Review.
-
Rosiglitazone promotes PPARγ-dependent and -independent alterations in gene expression in mouse islets.Endocrinology. 2012 Oct;153(10):4593-9. doi: 10.1210/en.2012-1243. Epub 2012 Jul 17. Endocrinology. 2012. PMID: 22807489 Free PMC article.
-
Effects of rosiglitazone on the configuration of action potentials and ion currents in canine ventricular cells.Br J Pharmacol. 2011 Jun;163(3):499-509. doi: 10.1111/j.1476-5381.2011.01215.x. Br J Pharmacol. 2011. PMID: 21232044 Free PMC article.
-
Thiazolidinediones (TZDs) enhance insulin secretory response via GPR40 and adenylate cyclase (AC).J Cell Physiol. 2021 Dec;236(12):8137-8147. doi: 10.1002/jcp.30467. Epub 2021 Jun 16. J Cell Physiol. 2021. PMID: 34133753 Free PMC article.
References
-
- AUGSTEIN P., DUNGER A., HEINKE P., WACHLIN G., BERG S., HEHMKE B., SALZSIEDER E. Prevention of autoimmune diabetes in NOD mice by troglitazone is associated with modulation of ICAM-1 expression on pancreatic islet cells and IFN-gamma expression in splenic T cells. Biochem. Biophys. Res. Commun. 2003;304:378–384. - PubMed
-
- BRAISSANT O., FOUFELLE F., SCOTTO C., DAUCA M., WAHLI W. Differential expression of peroxisome proliferator-activated receptors (PPARs): tissue distribution of PPAR-alpha, -beta, and -gamma in the adult rat. Endocrinology. 1996;137:354–366. - PubMed
-
- BUCHANAN T.A., XIANG A.H., PETERS R.K., KIOS S.L., MARROQUIN A., GOICO J., OCHOA C., TAN S., BERKOWITZ K., HODIS H.N., ZEN S.P. Preservation of pancreatic beta-cell function and prevention of type 2 diabetes by pharmacological treatment of insulin resistance in high-risk hispanic women. Diabetes. 2002;51:2796–2803. - PubMed
-
- BUCKINGHAM R.E., AL-BARAZANJI K.A., TOSELAND C.D., SLAUGHTER M., CONNOR S.C., WEST A., BOND B., TURNER N.C., CLAPHAM J.C. Peroxisome proliferator-activated receptor-gamma agonist, rosiglitazone, protects against nephropathy and pancreatic islet abnormalities in Zucker fatty rats. Diabetes. 1998;47:1326–1334. - PubMed
-
- CAMP H.S. Thiazolidinediones in diabetes: current status and future outlook. Curr. Opin. Investig. Drugs. 2003;4:406–411. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

