Ubiquitin-dependent lysosomal degradation of the HNE-modified proteins in lens epithelial cells
- PMID: 15247152
- PMCID: PMC1382276
- DOI: 10.1096/fj.04-1743fje
Ubiquitin-dependent lysosomal degradation of the HNE-modified proteins in lens epithelial cells
Abstract
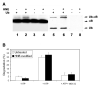
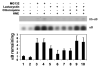
4-hydroxynonenal (HNE), a highly reactive lipid peroxidation product, may adversely modify proteins. Accumulation of HNE-modified proteins may be responsible for pathological lesions associated with oxidative stress. The objective of this work was to determine how HNE-modified proteins are removed from cells. The data showed that alphaB-crystallin modified by HNE was ubiquitinated at a faster rate than that of native alphaB-crystallin in a cell-free system. However, its susceptibility to proteasome-dependent degradation in the cell-free system did not increase. When delivered into cultured lens epithelial cells, HNE-modified alphaB-crystallin was degraded at a faster rate than that of unmodified alphaB-crystallin. Inhibition of the lysosomal activity stabilized HNE-modified alphaB-crystallin, but inhibition of the proteasome activity alone had little effect. To determine if other HNE-modified proteins are also degraded in a ubiquitin-dependent lysosomal pathway, lens epithelial cells were treated with HNE and the removal of HNE-modified proteins in the cells was monitored. The levels of HNE-modified proteins in the cell decreased rapidly upon removal of HNE from the medium. Depletion of ATP or the presence of MG132, a proteasome/lysosome inhibitor, resulted in stabilization of HNE-modified proteins. However, proteasome-specific inhibitors, lactacystin-beta-lactone and epoxomicin, could not stabilize HNE-modified proteins in the cells. In contrast, chloroquine, a lysosome inhibitor, stabilized HNE-modified proteins. The enrichment of HNE-modified proteins in the fraction of ubiquitin conjugates suggests that HNE-modified proteins are preferentially ubiquitinated. Taken together, these findings show that HNE-modified proteins are degraded via a novel ubiquitin and lysosomal-dependent but proteasome-independent pathway.
Figures





References
-
- Taylor A, Davies KLA. Protein oxidation and loss of protease activity may lead to cataract formation in the aged lens. Free Radic. Biol. Med. 1987;3:371–377. - PubMed
-
- Reddan JR, Sevilla MD, Giblin FJ, Padgaonkar V, Dziedzic DC, Leverenz V, Misra IC, Peters JL. The superoxide dismutase mimic TEMPOL protects culture rabbit lens epithelial cells from hydrogen peroxide insult. Exp. Eye Res. 1993;56:543–554. - PubMed
-
- Blondin J, Taylor A. Measures of leucine aminopeptidase can be used to anticipate UV-induced age-related damage to lens proteins: ascorbate can delay this damage. Mech. Ageing Dev. 1987;41:39–46. - PubMed
-
- Stadtman ER. Covalent modification reactions are marking steps in protein turnover. Biochemistry. 1990;29:6323–6331. - PubMed
-
- Windsor DP, White IG, Selley ML, Swan MA. Effects of the lipid peroxidation product (E)-4-hydroxy-2-nonenal on ram sperm function. J. Reprod. Fertil. 1993;99:359–366. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

