Inhibition of macrophage migration inhibitory factor decreases proliferation and cytokine expression in bladder cancer cells
- PMID: 15248897
- PMCID: PMC481073
- DOI: 10.1186/1471-2407-4-34
Inhibition of macrophage migration inhibitory factor decreases proliferation and cytokine expression in bladder cancer cells
Abstract
Background: The importance of various inflammatory cytokines in maintaining tumor cell growth and viability is well established. Increased expression of the proinflammatory cytokine macrophage migration inhibitory factor (MIF) has previously been associated with various types of adenocarcinoma.
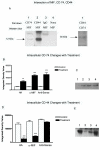
Methods: MIF IHC was used to localize MIF in human bladder tissue. ELISA and Western blot analysis determined the synthesis and secretion of MIF by human bladder transitional cell carcinoma cells. The effects of MIF inhibitors (high molecular weight hyaluronate (HA), anti-MIF antibody or MIF anti-sense) on cell growth and cytokine expression were analyzed.
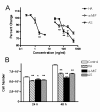
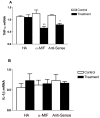
Results: Human bladder cancer cells (HT-1376) secrete detectable amounts of MIF protein. Treatment with HA, anti-MIF antibody and MIF anti-sense reduced HT-1376 cell proliferation, MIF protein secretion, MIF gene expression and secreted inflammatory cytokines. Our evidence suggests MIF interacts with the invariant chain, CD74 and the major cell surface receptor for HA, CD44.
Conclusions: This study is the first to report MIF expression in the human bladder and these findings support a role for MIF in tumor cell proliferation. Since MIF participates in the inflammatory response and bladder cancer is associated with chronic inflammatory conditions, these new findings suggest that neutralizing bladder tumor MIF may serve as a novel therapeutic treatment for bladder carcinoma.
Figures

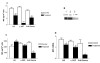
References
-
- Jemal A, Murray T, Samuels A, Ghafoor A, Ward E, Thun MJ. Cancer statistics. CA Cancer J Clin. 2003;53:5–26. - PubMed
-
- Piazza GA, Thompson WJ, Pamukeu R, Alila HW, Whitehead CM, Liu L, Fetter JR, Gresh WE, Jr, Klein-Szanto AJ, Farnell DR, Eto I, Grubbs CJ. Exisulind, a novel proapoptotic drug, inhibits rat urinary bladder tumorigenesis. Cancer Res. 2001;61:3961–3968. - PubMed
-
- Herr HW, Badalament RA, Amato DA, Laudone VP, Fair WR, Whitmore WF., Jr Superficial bladder cancer treated with bacillus Calmette-Guerin: a multivariate analysis of factors affecting tumor progression. J Urol. 1989;141:22–29. - PubMed
-
- La Vecchia C, Negri E, D'Avanzo B, Savoldelli R, Francesci S. Genital and urinary tract disease and bladder cancer. Cancer Res. 1991;51:629–631. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

