Cortactin and Crk cooperate to trigger actin polymerization during Shigella invasion of epithelial cells
- PMID: 15263018
- PMCID: PMC2172305
- DOI: 10.1083/jcb.200402073
Cortactin and Crk cooperate to trigger actin polymerization during Shigella invasion of epithelial cells
Abstract
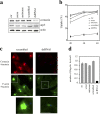
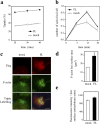
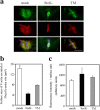
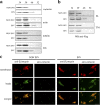
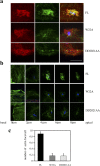
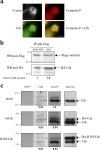
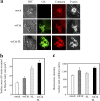
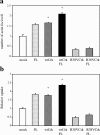
Shigella, the causative agent of bacillary dysentery, invades epithelial cells in a process involving Src tyrosine kinase signaling. Cortactin, a ubiquitous actin-binding protein present in structures of dynamic actin assembly, is the major protein tyrosine phosphorylated during Shigella invasion. Here, we report that RNA interference silencing of cortactin expression, as does Src inhibition in cells expressing kinase-inactive Src, interferes with actin polymerization required for the formation of cellular extensions engulfing the bacteria. Shigella invasion induced the recruitment of cortactin at plasma membranes in a tyrosine phosphorylation-dependent manner. Overexpression of wild-type forms of cortactin or the adaptor protein Crk favored Shigella uptake, and Arp2/3 binding-deficient cortactin derivatives or an Src homology 2 domain Crk mutant interfered with bacterial-induced actin foci formation. Crk was shown to directly interact with tyrosine-phosphorylated cortactin and to condition cortactin-dependent actin polymerization required for Shigella uptake. These results point at a major role for a Crk-cortactin complex in actin polymerization downstream of tyrosine kinase signaling.
Copyright The Rockerfeller University Press
Figures
References
-
- Bowden, E.T., M. Barth, D. Thomas, R.I. Glazer, and S.C. Mueller. 1999. An invasion-related complex of cortactin, paxillin and PKCμ associates with invadopodia at sites of extracellular matrix degradation. Oncogene. 18:4440–4449. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

