Ancient adaptive evolution of the primate antiviral DNA-editing enzyme APOBEC3G
- PMID: 15269786
- PMCID: PMC479043
- DOI: 10.1371/journal.pbio.0020275
Ancient adaptive evolution of the primate antiviral DNA-editing enzyme APOBEC3G
Abstract
Host genomes have adopted several strategies to curb the proliferation of transposable elements and viruses. A recently discovered novel primate defense against retroviral infection involves a single-stranded DNA-editing enzyme, APOBEC3G, that causes hypermutation of HIV. The HIV-encoded virion infectivity factor (Vif) protein targets APOBEC3G for destruction, setting up a genetic conflict between the APOBEC3G and Vif genes. This kind of conflict leads to rapid fixation of mutations that alter amino acids at the protein-protein interface, referred to as positive selection. We show that the APOBEC3G gene has been subject to strong positive selection throughout the history of primate evolution. Unexpectedly, this selection appears more ancient than, and is likely only partially caused by, modern lentiviruses. Furthermore, five additional APOBEC genes in the human genome appear to be engaged in similar genetic conflicts, displaying some of the highest signals for positive selection in the human genome. Despite being only recently discovered, editing of RNA and DNA may thus represent an ancient form of host defense in primate genomes.
Conflict of interest statement
The authors have declared that no conflicts of interest exist.
Figures
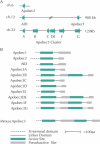



References
-
- Anisimova M, Bielawski JP, Yang Z. Accuracy and power of Bayes prediction of amino acid sites under positive selection. Mol Biol Evol. 2002;19:950–958. - PubMed
-
- Chan L, Chang BHJ, Nakamuta M, Li WH, Smith LC. Apobec-1 and apolipoprotein B mRNA editing. Biochim Biophys Acta. 1997;1345:11–26. - PubMed
-
- Comeron JM. K-estimator: Calculation of the number of nucleotide substitutions per site and the confidence intervals. Bioinformatics. 1999;15:763–764. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

