Subfertility and defective folliculogenesis in female mice lacking androgen receptor
- PMID: 15277682
- PMCID: PMC509185
- DOI: 10.1073/pnas.0404372101
Subfertility and defective folliculogenesis in female mice lacking androgen receptor
Abstract
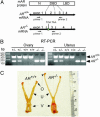
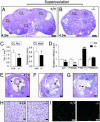
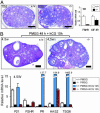
The roles of the androgen receptor (AR) in female fertility and ovarian function remain largely unknown. Here we report on the generation of female mice lacking AR (AR(-/-)) and the resulting influences on the reproductive system. Female AR(-/-) mice appear normal but show longer estrous cycles and reduced fertility. The ovaries from sexually mature AR(-/-) females exhibited a marked reduction in the number of corpora lutea. After superovulation treatment, the AR(-/-) ovaries produced fewer oocytes and also showed fewer corpora lutea. During the periovulatory period, an intensive granulosa apoptosis event occurs in the AR(-/-) preovulatory follicles, concurrent with the down-regulation of p21 and progesterone receptor expression. Furthermore, the defective conformation of the cumulus cell-oocyte complex from the AR(-/-) females implies a lower fertilization capability of the AR(-/-) oocytes. In addition to insufficient progesterone production, the diminished endometrial growth in uteri in response to exogenous gonadotropins indicates that AR(-/-) females exhibit a luteal phase defect. Taken together, these data provide in vivo evidence showing that AR plays an important role in female reproduction.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

