Nitric oxide as an endogenous mutagen for Sendai virus without antiviral activity
- PMID: 15280479
- PMCID: PMC479088
- DOI: 10.1128/JVI.78.16.8709-8719.2004
Nitric oxide as an endogenous mutagen for Sendai virus without antiviral activity
Abstract
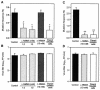
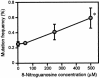
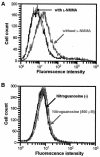
Nitric oxide (NO) may affect the genomes of various pathogens, and this mutagenesis is of particular interest for viral pathogenesis and evolution. Here, we investigated the effect of NO on viral replication and mutation. Exogenous or endogenous NO had no apparent antiviral effect on influenza A virus and Sendai virus. The mutagenic potential of NO was analyzed with Sendai virus fused to a green fluorescent protein (GFP) gene (GFP-SeV). GFP-SeV was cultured in SW480 cells transfected with a vector expressing inducible NO synthase (iNOS). The mutation frequency of GFP-SeV was examined by measuring loss of GFP fluorescence of the viral plaques. GFP-SeV mutation frequency in iNOS-SW480 cells was much higher than that in parent SW480 cells and was reduced to the level of mutation frequency in the parent cells by treatment with an NO synthase (NOS) inhibitor. Immunocytochemistry showed generation of more 8-nitroguanosine in iNOS-SW480 cells than in SW480 cells without iNOS transfection. Authentic 8-nitroguanosine added exogenously to GFP-SeV-infected CV-1 cells increased the viral mutation frequency. Profiles of the GFP gene mutations induced by 8-nitroguanosine appeared to resemble those of mutations occurring in mouse lungs in vivo. A base substitution that was characteristic of both mutants (those induced by 8-nitroguanosine and those occurring in vivo) was a C-to-U transition. NO-dependent oxidative stress in iNOS-SW480 cells was also evident. Together, the results indicate unambiguously that NO has mutagenic potential for RNA viruses such as Sendai virus without affecting viral replication, possibly via 8-nitroguanosine formation and cellular oxidative stress.
Figures



Similar articles
-
Viral mutation accelerated by nitric oxide production during infection in vivo.FASEB J. 2000 Jul;14(10):1447-54. doi: 10.1096/fj.14.10.1447. FASEB J. 2000. PMID: 10877838
-
8-nitroguanosine formation in viral pneumonia and its implication for pathogenesis.Proc Natl Acad Sci U S A. 2003 Jan 21;100(2):685-90. doi: 10.1073/pnas.0235623100. Epub 2003 Jan 8. Proc Natl Acad Sci U S A. 2003. PMID: 12522148 Free PMC article.
-
Sendai virus C protein inhibits lipopolysaccharide-induced nitric oxide production through impairing interferon-β signaling.Int Immunopharmacol. 2014 Nov;23(1):267-72. doi: 10.1016/j.intimp.2014.09.012. Epub 2014 Sep 19. Int Immunopharmacol. 2014. PMID: 25242386
-
Nitrative stress through formation of 8-nitroguanosine: insights into microbial pathogenesis.Nitric Oxide. 2006 Mar;14(2):101-8. doi: 10.1016/j.niox.2005.10.004. Epub 2005 Nov 23. Nitric Oxide. 2006. PMID: 16309933 Review.
-
Inducible nitric oxide synthase in the liver: regulation and function.Biochemistry (Mosc). 1998 Jul;63(7):766-81. Biochemistry (Mosc). 1998. PMID: 9721329 Review.
Cited by
-
Visualizing inducible nitric-oxide synthase in living cells with a heme-binding fluorescent inhibitor.Proc Natl Acad Sci U S A. 2005 Jul 19;102(29):10117-22. doi: 10.1073/pnas.0408972102. Epub 2005 Jul 8. Proc Natl Acad Sci U S A. 2005. PMID: 16006534 Free PMC article.
-
The not-so-infinite malleability of RNA viruses: Viral and cellular determinants of RNA virus mutation rates.PLoS Pathog. 2017 Apr 27;13(4):e1006254. doi: 10.1371/journal.ppat.1006254. eCollection 2017 Apr. PLoS Pathog. 2017. PMID: 28448634 Free PMC article. Review. No abstract available.
-
Sendai virus intra-host population dynamics and host immunocompetence influence viral virulence during in vivo passage.Virus Evol. 2016 Apr 9;2(1):vew008. doi: 10.1093/ve/vew008. eCollection 2016 Jan. Virus Evol. 2016. PMID: 27774301 Free PMC article.
-
Nitric oxide in dengue and dengue haemorrhagic fever: necessity or nuisance?FEMS Immunol Med Microbiol. 2009 Jun;56(1):9-24. doi: 10.1111/j.1574-695X.2009.00544.x. Epub 2009 Feb 23. FEMS Immunol Med Microbiol. 2009. PMID: 19239490 Free PMC article. Review.
-
An aged host promotes the evolution of avirulent coxsackievirus into a virulent strain.Proc Natl Acad Sci U S A. 2006 Sep 12;103(37):13825-30. doi: 10.1073/pnas.0605507103. Epub 2006 Sep 1. Proc Natl Acad Sci U S A. 2006. PMID: 16950876 Free PMC article.
References
-
- Adachi, H., S. Iida, S. Oguchi, H. Ohshima, H. Suzuki, K. Nagasaki, H. Kawasaki, T. Sugimura, and H. Esumi. 1993. Molecular cloning of a cDNA encoding an inducible calmodulin-dependent nitric-oxide synthase from rat liver and its expression in COS 1 cells. Eur. J. Biochem. 217:37-43. - PubMed
-
- Akaike, T., S. Fujii, A. Kato, J. Yoshitake, Y. Miyamoto, T. Sawa, S. Okamoto, M. Suga, M. Asakawa, Y. Nagai, and H. Maeda. 2000. Viral mutation accelerated by nitric oxide production during infection in vivo. FASEB J. 14:1447-1454. - PubMed
-
- Akaike, T., K. Inoue, T. Okamoto, H. Nishino, M. Otagiri, S. Fujii, and H. Maeda. 1997. Nanomolar quantification and identification of various nitrosothiols by high performance liquid chromatography coupled with flow reactors of metals and Griess reagent. J. Biochem. 122:459-466. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

