Porcine endogenous retroviruses infect cells lacking cognate receptors by an alternative pathway: implications for retrovirus evolution and xenotransplantation
- PMID: 15280495
- PMCID: PMC479092
- DOI: 10.1128/JVI.78.16.8868-8877.2004
Porcine endogenous retroviruses infect cells lacking cognate receptors by an alternative pathway: implications for retrovirus evolution and xenotransplantation
Abstract
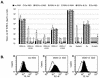
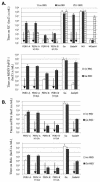
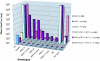
A PHQ motif near the amino termini of gammaretroviral envelope glycoprotein surface (SU) subunits is important for infectivity but not for incorporation into virions or binding to cognate receptors. The H residue of this motif is most critical, with all substitutions we tested being inactive. Interestingly, porcine endogenous retroviruses (PERVs) of all three host-range groups, A, B, and C, lack full PHQ motifs, but most members have an H residue at position 10. H10A PERV mutants are noninfectious but were efficiently transactivated by adding to the assays a PHQ-containing SU or receptor-binding subdomain (RBD) derived from a gibbon ape leukemia virus (GALV). A requirement of this transactivation was a functional GALV receptor on the cells. In contrast to this heterologous transactivation, PERV RBDs and SUs were inactive in all tested cells, including porcine ST-IOWA cells. Surprisingly, transactivation by GALV RBD enabled wild-type or H10A mutant PERVs of all three host-range groups to efficiently infect cells from humans and rodents that lack functional PERV receptors and it substantially enhanced infectivities of wild-type PERVs, even for cells with PERV receptors. Thus, PERVs can suboptimally infect cells that contain cognate receptors or they can employ a transactivation pathway to more efficiently infect all cells. This ability to infect cells lacking cognate receptors was previously demonstrated only for nontransmissible variant gammaretroviruses with recombinant and mutant envelope glycoproteins. We conclude that some endogenously inherited mammalian retroviruses also have a receptor-independent means for overcoming host-range and interference barriers, implying a need for caution in xenotransplantation, especially of porcine tissues.
Figures




References
-
- Anderson, M. M., A. S. Lauring, C. C. Burns, and J. Overbaugh. 2000. Identification of a cellular cofactor required for infection by feline leukemia virus. Science 287:1828-1830. - PubMed
-
- Babe, L. M., A. Unal, and C. S. Craik. 1997. Obstruction of HIV-1 particle release by interferon-alpha occurs before viral protease processing and is independent of envelope glycoprotein. J. Interferon Cytokine Res. 17:287-293. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

