Insufficient folding of type IV collagen and formation of abnormal basement membrane-like structure in embryoid bodies derived from Hsp47-null embryonic stem cells
- PMID: 15282337
- PMCID: PMC519141
- DOI: 10.1091/mbc.e04-01-0050
Insufficient folding of type IV collagen and formation of abnormal basement membrane-like structure in embryoid bodies derived from Hsp47-null embryonic stem cells
Abstract
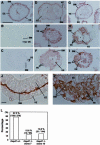
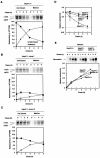
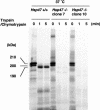
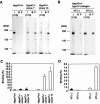
Hsp47 is a molecular chaperone that specifically recognizes procollagen in the endoplasmic reticulum. Hsp47-null mouse embryos produce immature type I collagen and form discontinuous basement membranes. We established Hsp47-/- embryonic stem cell lines and examined formation of basement membrane and production of type IV collagen in embryoid bodies, a model for postimplantation egg-cylinder stage embryos. The visceral endodermal cell layers surrounding Hsp47-/- embryoid bodies were often disorganized, a result that suggested abnormal function of the basement membrane under the visceral endoderm. Rate of type IV collagen secretion by Hsp47-/- cells was fourfold lower than that of Hsp47+/+ cells. Furthermore, type IV collagen secreted from Hsp47-/- cells was much more sensitive to protease digestion than was type IV collagen secreted from Hsp47+/+ cells, which suggested insufficient or incorrect triple helix formation in type IV collagen in the absence of Hsp47. These results indicate for the first time that Hsp47 is required for the molecular maturation of type IV collagen and suggest that misfolded type IV collagen causes abnormal morphology of embryoid bodies.
Figures

Similar articles
-
Accumulation of type IV collagen in dilated ER leads to apoptosis in Hsp47-knockout mouse embryos via induction of CHOP.J Cell Sci. 2004 Nov 15;117(Pt 24):5913-22. doi: 10.1242/jcs.01514. Epub 2004 Nov 2. J Cell Sci. 2004. PMID: 15522896
-
Roles of collagen fibers and its specific molecular chaperone: analysis using HSP47-knockout mice.Biol Sci Space. 2004 Nov;18(3):118-9. Biol Sci Space. 2004. PMID: 15858349
-
HSP47 as a collagen-specific molecular chaperone: function and expression in normal mouse development.Semin Cell Dev Biol. 2003 Oct;14(5):275-82. doi: 10.1016/j.semcdb.2003.09.020. Semin Cell Dev Biol. 2003. PMID: 14986857 Review.
-
Type I collagen in Hsp47-null cells is aggregated in endoplasmic reticulum and deficient in N-propeptide processing and fibrillogenesis.Mol Biol Cell. 2006 May;17(5):2346-55. doi: 10.1091/mbc.e05-11-1065. Epub 2006 Mar 8. Mol Biol Cell. 2006. PMID: 16525016 Free PMC article.
-
[Therapeutic strategy for fibrotic diseases by regulating the expression of collagen-specific molecular chaperone HSP47].Nihon Yakurigaku Zasshi. 2003 Jan;121(1):4-14. doi: 10.1254/fpj.121.4. Nihon Yakurigaku Zasshi. 2003. PMID: 12617032 Review. Japanese.
Cited by
-
Beyond classical collagen: basement membrane collagen IV in age-associated lung diseases.Eur Respir Rev. 2025 Jul 23;34(177):240192. doi: 10.1183/16000617.0192-2024. Print 2025 Jul. Eur Respir Rev. 2025. PMID: 40701642 Free PMC article. Review.
-
Direct in vitro and in vivo evidence for interaction between Hsp47 protein and collagen triple helix.J Biol Chem. 2012 Feb 24;287(9):6810-8. doi: 10.1074/jbc.M111.280248. Epub 2012 Jan 10. J Biol Chem. 2012. PMID: 22235129 Free PMC article.
-
Decreased Expression of Heat Shock Protein 47 Is Associated with T-2 Toxin and Low Selenium-Induced Matrix Degradation in Cartilages of Kashin-Beck Disease.Biol Trace Elem Res. 2021 Mar;199(3):944-954. doi: 10.1007/s12011-020-02237-1. Epub 2020 Jun 26. Biol Trace Elem Res. 2021. PMID: 32591934
-
Deletion of the collagen-specific molecular chaperone Hsp47 causes endoplasmic reticulum stress-mediated apoptosis of hepatic stellate cells.J Biol Chem. 2015 Feb 6;290(6):3639-46. doi: 10.1074/jbc.M114.592139. Epub 2014 Dec 18. J Biol Chem. 2015. PMID: 25525267 Free PMC article.
-
HSP47 Increases the Expression of Type I Collagen in Fibroblasts through IRE1α Activation, XBP1 Splicing, and Nuclear Translocation of β-Catenin.Cells. 2024 Mar 17;13(6):527. doi: 10.3390/cells13060527. Cells. 2024. PMID: 38534372 Free PMC article.
References
-
- Bachinger, H.P., Bruckner, P., Timpl, R., Prockop, D.J., and Engel, J. (1980). Folding mechanism of the triple helix in type-III collagen and type-III pN-collagen. Role of disulfide bridges and peptide bond isomerization. Eur. J. Biochem. 106, 619-632. - PubMed
-
- Berg, R.A., P.D. (1973). The thermal transition of a non-hydroxylated form of collagen. Evidence for a role for hydroxyproline in stabilizing the triple-helix of collagen. Biochem. Biophys. Res. Commun. 52, 115-120. - PubMed
-
- Bottomley, M.J., Batten, M.R., Lumb, R.A., and Bulleid, N.J. (2001). Quality control in the endoplasmic reticulum: PDI mediates the ER retention of unassembled procollagen C-propeptides. Curr. Biol. 11, 1114-1118. - PubMed
-
- Bradford, M.M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248-254. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

