The LC7 light chains of Chlamydomonas flagellar dyneins interact with components required for both motor assembly and regulation
- PMID: 15304520
- PMCID: PMC519155
- DOI: 10.1091/mbc.e04-06-0461
The LC7 light chains of Chlamydomonas flagellar dyneins interact with components required for both motor assembly and regulation
Abstract
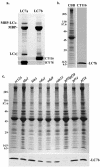
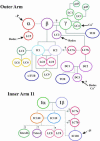
Members of the LC7/Roadblock family of light chains (LCs) have been found in both cytoplasmic and axonemal dyneins. LC7a was originally identified within Chlamydomonas outer arm dynein and associates with this motor's cargo-binding region. We describe here a novel member of this protein family, termed LC7b that is also present in the Chlamydomonas flagellum. Levels of LC7b are reduced approximately 20% in axonemes isolated from strains lacking inner arm I1 and are approximately 80% lower in the absence of the outer arms. When both dyneins are missing, LC7b levels are diminished to <10%. In oda9 axonemal extracts that completely lack outer arms, LC7b copurifies with inner arm I1, whereas in ida1 extracts that are devoid of I1 inner arms it associates with outer arm dynein. We also have observed that some LC7a is present in both isolated axonemes and purified 18S dynein from oda1, suggesting that it is also a component of both the outer arm and inner arm I1. Intriguingly, in axonemal extracts from the LC7a null mutant, oda15, which assembles approximately 30% of its outer arms, LC7b fails to copurify with either dynein, suggesting that it interacts with LC7a. Furthermore, both the outer arm gamma heavy chain and DC2 from the outer arm docking complex completely dissociate after salt extraction from oda15 axonemes. EDC cross-linking of purified dynein revealed that LC7b interacts with LC3, an outer dynein arm thioredoxin; DC2, an outer arm docking complex component; and also with the phosphoprotein IC138 from inner arm I1. These data suggest that LC7a stabilizes both the outer arms and inner arm I1 and that both LC7a and LC7b are involved in multiple intradynein interactions within both dyneins.
Figures








References
-
- Benashski, S.E., and King, S.M. (2000). Investigation of protein-protein interactions within flagellar dynein using homobifunctional and zero-length crosslinking reagents. Methods 22, 365-371. - PubMed
-
- Benashski, S.E., Patel-King, R.S., and King, S.M. (1999). Light chain 1 from the Chlamydomonas outer dynein arm is a leucine-rich repeat protein associated with the motor domain of the γ heavy chain. Biochemistry 38, 7253-7264. - PubMed
-
- Bowman, A.B., Patel-King, R.S., Benashski, S.E., McCaffery, J.M., Goldstein, L.S., and King, S.M. (1999). Drosophila roadblock and Chlamydomonas LC 7, a conserved family of dynein-associated proteins involved in axonal transport, flagellar motility, and mitosis. J. Cell Biol. 146, 165-180. - PMC - PubMed
-
- Casey, D.M., Inaba, K., Pazour, G.J., Takada, S., Wakabayashi, K., Wilkerson, C.G., Kamiya, R., and Witman, G.B. (2003). DC3, the 21-kDa subunit of the outer dynein arm-docking complex (ODA-DC), is a novel EF-hand protein important for assembly of both the outer arm and the ODA-DC. Mol. Biol. Cell 14, 3650-3663. - PMC - PubMed
-
- DiBella, L.M., Benashski, S.E., Tedford, H.W., Harrison, A., Patel-King, R.S., and King, S.M. (2001). The Tctex1/Tctex2 class of dynein light chains: dimerization, differential expression, and interaction with the LC8 protein family. J. Biol. Chem. 276, 14366-14373. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

