Matrix metalloproteinase-2 cleavage of adrenomedullin produces a vasoconstrictor out of a vasodilator
- PMID: 15307819
- PMCID: PMC1133733
- DOI: 10.1042/BJ20040920
Matrix metalloproteinase-2 cleavage of adrenomedullin produces a vasoconstrictor out of a vasodilator
Abstract
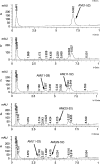
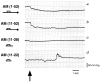
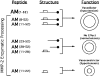
MMPs (matrix metalloproteinases) play a major role in the pathogenesis of hypertension by altering the extracellular matrix during cardiovascular remodelling. In the present study we show that MMP-2, but not MMP-9, cleaves the vasodilator peptide AM (adrenomedullin). Addition of the AM-binding protein, complement factor H, prevents this cleavage, providing a hitherto unknown mechanism of action for this binding protein. We identified the signature cleavage fragments and found some of them in human urine, suggesting that MMP-2 processing of AM may occur in vivo. Synthetic AM fragments regulated blood pressure in rats. The larger peptides are vasodilators, as is intact AM, whereas intermediate fragments did not affect blood pressure. In contrast, AM(11-22) elicited vasoconstriction. Studies of AM receptor activation in Rat2 cells confirm that the larger AM cleavage peptides activated this receptor, whereas AM(11-22) did not. The present study defines a new mechanism through which MMP-2 may regulate blood pressure by simultaneously eliminating a vasodilator and generating a vasoconstrictor.
Figures



Comment in
-
Dilating the degradome: matrix metalloproteinase 2 (MMP-2) cuts to the heart of the matter.Biochem J. 2004 Nov 1;383(Pt. 3):e5-7. doi: 10.1042/bj20041433. Biochem J. 2004. PMID: 15508185 Free PMC article. Review.
References
-
- Yildirir A., Batur M. K., Oto A. Hypertension and arrhytmia: blood pressure control and beyond. Europace. 2002;4:175–182. - PubMed
-
- Mulvany M. J. Small artery remodeling and significance in the development of hypertension. News Physiol. Sci. 2002;17:105–109. - PubMed
-
- D'Armiento J. Matrix metalloproteinase disruption of the extracellular matrix and cardiac dysfunction. Trends Cardiovasc. Med. 2002;12:97–101. - PubMed
-
- Intengan H. D., Schiffrin E. L. Vasopeptidase inhibition has potent effects on blood pressure and resistance arteries in stroke-prone spontaneously hypertensive rats. Hypertension. 2001;38:581–587. - PubMed
-
- Brinckerhoff C. E., Matrisian L. M. Matrix metalloproteinases: a tail of a frog that became a prince. Nat. Rev. Mol. Cell Biol. 2002;3:207–214. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

