Thymocytes induced by antigen injection into the anterior chamber activate splenic CD8+ suppressor cells and enhance the antigen-induced production of immunoglobulin G1 antibodies
- PMID: 15312135
- PMCID: PMC1782546
- DOI: 10.1111/j.1365-2567.2004.01928.x
Thymocytes induced by antigen injection into the anterior chamber activate splenic CD8+ suppressor cells and enhance the antigen-induced production of immunoglobulin G1 antibodies
Abstract
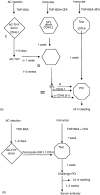
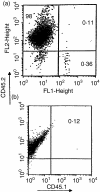
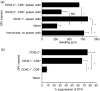
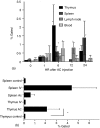
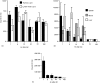
Injection of antigen into the ocular anterior chamber (AC) of a mouse eye (an immunologically privileged site) induces the activation of immunoregulatory NK1.1+, CD4- CD8-, T-cell receptor (TCR) alphabeta+ thymocytes. These thymocytes transfer the suppression of delayed-type hypersensitivity (DTH) when injected into mice sensitized to the same antigen but do not effect the suppression of DTH. On the other hand, the immunized recipients of these transferred thymocytes produce splenic CD8+ T cells that effect the suppression of DTH. However, it is unclear whether the thymocytes transferred from the AC-injected donor differentiate into and/or activate CD8+ T-splenic suppressor cells. We therefore sought to determine the origin of splenic suppressor cells produced in the recipients of immunoregulatory thymocytes transferred from donors that receive an injection of antigen into the AC. CD45.1+ thymocytes from mice that received an AC injection of 2,4,6-trinitrobenzene sulphonic acid (TNP)-bovine serum albumin (BSA) were transferred to congenic CD45.2+ TNP-BSA-immune recipients. Spleen cells from the recipients were then sorted based on anti-CD45.1 or -CD45.2 antibody binding and assayed for suppressor cells. This was done by the injection of separated spleen cells into the footpad of TNP-BSA-immunized mice, concurrent with the induction of footpad swelling (contact sensitivity) of the footpad elicited by an epicutaneous application of picryl chloride. The systemic distribution of antigen after the injection of antigen into the AC was demonstrated by the injection of fluorescein or 125I-labelled TNP-BSA into the AC. The results demonstrate that (i) splenic CD8+ T-suppressor cells produced in the immunized recipients of immunoregulatory thymocytes are derived from the CD45.2 recipient of the CD45.1+ thymocytes; (ii) the induction of recipient splenic suppressor T cells by the transferred immunoregulatory thymocytes requires that the recipient be immunized to the same antigen as that used to induce immunoregulatory thymocytes; (iii) antigen is introduced to the thymus after an injection of antigen into the AC; (iv) although the transfer of the suppression of DTH by regulatory thymocytes was not dependent on interleukin-4 (IL-4), CD4+ NK1.1- regulatory thymocytes from AC-injected donors enhanced the production of immunoglobulin G1 antibodies to TNP-BSA by an IL-4-dependent mechanism. These observations suggest that the adult thymus plays an active role in the induction and maintenance of anterior chamber-associated immune deviation as manifested by the generation of the suppression of cell-mediated immunity to exogenous antigen and the antigen-induced production of IgG1 antibodies.
Figures



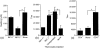

Similar articles
-
The suppression of delayed-type hypersensitivity by CD8+ regulatory T cells requires interferon-gamma.Immunology. 2007 Jan;120(1):112-9. doi: 10.1111/j.1365-2567.2006.02486.x. Epub 2006 Oct 18. Immunology. 2007. PMID: 17052246 Free PMC article.
-
Phenotypic and immunoregulatory characteristics of monocytic iris cells.Immunology. 2006 Apr;117(4):566-75. doi: 10.1111/j.1365-2567.2006.02333.x. Immunology. 2006. PMID: 16556271 Free PMC article.
-
The induction of splenic suppressor T cells through an immune-privileged site requires an intact sympathetic nervous system.J Neuroimmunol. 2004 Aug;153(1-2):40-9. doi: 10.1016/j.jneuroim.2004.04.008. J Neuroimmunol. 2004. PMID: 15265662
-
Control of delayed-type hypersensitivity by ocular- induced CD8+ regulatory t cells.Chem Immunol Allergy. 2008;94:138-149. doi: 10.1159/000154998. Chem Immunol Allergy. 2008. PMID: 18802344 Review.
-
Mechanisms of mouse T lymphocyte-induced suppression of the IgG2ab allotype and T lymphocyte tolerance to IgG2ab.Arch Immunol Ther Exp (Warsz). 2001;49(6):407-15. Arch Immunol Ther Exp (Warsz). 2001. PMID: 11814234 Review.
Cited by
-
Transfer of Maternal Immune Cells by Breastfeeding: Maternal Cytotoxic T Lymphocytes Present in Breast Milk Localize in the Peyer's Patches of the Nursed Infant.PLoS One. 2016 Jun 10;11(6):e0156762. doi: 10.1371/journal.pone.0156762. eCollection 2016. PLoS One. 2016. PMID: 27285085 Free PMC article.
-
Immune amplification of murine CD8 suppressor T cells induced via an immune-privileged site: quantifying suppressor T cells functionally.PLoS One. 2011;6(8):e22496. doi: 10.1371/journal.pone.0022496. Epub 2011 Aug 2. PLoS One. 2011. PMID: 21829628 Free PMC article.
-
T cell sensitivity to TGF-beta is required for the effector function but not the generation of splenic CD8+ regulatory T cells induced via the injection of antigen into the anterior chamber.Int Immunol. 2009 May;21(5):567-74. doi: 10.1093/intimm/dxp023. Epub 2009 Mar 26. Int Immunol. 2009. PMID: 19325036 Free PMC article.
-
The suppression of delayed-type hypersensitivity by CD8+ regulatory T cells requires interferon-gamma.Immunology. 2007 Jan;120(1):112-9. doi: 10.1111/j.1365-2567.2006.02486.x. Epub 2006 Oct 18. Immunology. 2007. PMID: 17052246 Free PMC article.
-
A caveat for T cell transfer studies: generation of cytotoxic anti-Thy1.2 antibodies in Thy1.1 congenic mice given Thy1.2+ tumors or T cells.J Leukoc Biol. 2011 Feb;89(2):291-300. doi: 10.1189/jlb.0610333. Epub 2010 Oct 19. J Leukoc Biol. 2011. PMID: 20959413 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
