Farnesol is glucuronidated in human liver, kidney and intestine in vitro, and is a novel substrate for UGT2B7 and UGT1A1
- PMID: 15320866
- PMCID: PMC1134150
- DOI: 10.1042/BJ20040997
Farnesol is glucuronidated in human liver, kidney and intestine in vitro, and is a novel substrate for UGT2B7 and UGT1A1
Abstract
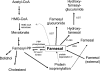
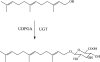
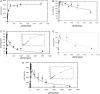
Farnesol is an isoprenoid found in many aromatic plants and is also produced in humans, where it acts on numerous nuclear receptors and has received considerable attention due to its apparent anticancer properties. Although farnesol has been studied for over 30 years, its metabolism has not been well characterized. Recently, farnesol was shown to be metabolized by cytochromes P450 in rabbit; however, neither farnesol hydroxylation nor glucuronidation in humans have been reported to date. In the present paper, we show for the first time that farnesol is metabolized to farnesyl glucuronide, hydroxyfarnesol and hydroxyfarnesyl glucuronide by human tissue microsomes, and we identify the specific human UGTs (uridine diphosphoglucuronosyltransferases) involved. Farnesol metabolism was examined by a sensitive LC (liquid chromatography)-MS/MS method. Results indicate that farnesol is a good substrate for glucuronidation in human liver, kidney and intestine microsomes (values in nmol/min per mg). Initial analysis using expressed human UGTs indicated that UGTs 1A1 and 2B7 were primarily responsible for glucuronidation in vitro, with significantly lower activity for all the other UGTs tested (UGTs 1A3, 1A4, 1A6, 1A9 and 2B4). Kinetic analysis and inhibition experiments indicate that, in liver microsomes, UGT1A1 is primarily responsible for farnesol glucuronidation; however, in intestine microsomes, UGT2B7 is probably the major isoform involved, with a very-low-micromolar K(m). We also show the first direct evidence that farnesol can be metabolized to hydroxyfarnesol by human liver microsomes and that hydroxyfarnesol is metabolized further to hydroxyfarnesyl glucuronide. Thus glucuronidation may modulate the physiological and/or pharmacological properties of this potent signalling molecule.
Figures



Similar articles
-
Characterization of UDP-glucuronosyltransferases involved in glucuronidation of diethylstilbestrol in human liver and intestine.Chem Res Toxicol. 2012 Dec 17;25(12):2663-9. doi: 10.1021/tx300310k. Epub 2012 Nov 13. Chem Res Toxicol. 2012. PMID: 23126256
-
Identification of human hepatic UDP-glucuronosyltransferases involved in the glucuronidation of temazepam.Drug Metab Dispos. 2025 Jul;53(7):100104. doi: 10.1016/j.dmd.2025.100104. Epub 2025 May 26. Drug Metab Dispos. 2025. PMID: 40543327
-
Epirubicin glucuronidation is catalyzed by human UDP-glucuronosyltransferase 2B7.Drug Metab Dispos. 2001 May;29(5):686-92. Drug Metab Dispos. 2001. PMID: 11302935
-
Renal drug metabolism in humans: the potential for drug-endobiotic interactions involving cytochrome P450 (CYP) and UDP-glucuronosyltransferase (UGT).Br J Clin Pharmacol. 2013 Oct;76(4):587-602. doi: 10.1111/bcp.12086. Br J Clin Pharmacol. 2013. PMID: 23362865 Free PMC article. Review.
-
Coordinate regulation of drug metabolism by xenobiotic nuclear receptors: UGTs acting together with CYPs and glucuronide transporters.Drug Metab Rev. 2004 Oct;36(3-4):595-615. doi: 10.1081/dmr-200033455. Drug Metab Rev. 2004. PMID: 15554238 Review.
Cited by
-
Acyclic sesquiterpenes nerolidol and farnesol: mechanistic insights into their neuroprotective potential.Pharmacol Rep. 2025 Feb;77(1):31-42. doi: 10.1007/s43440-024-00672-8. Epub 2024 Oct 22. Pharmacol Rep. 2025. PMID: 39436564 Review.
-
Functional characterization of OR51B5 and OR1G1 in human lung epithelial cells as potential drug targets for non-type 2 lung diseases.Cell Biol Toxicol. 2024 Nov 13;40(1):96. doi: 10.1007/s10565-024-09935-9. Cell Biol Toxicol. 2024. PMID: 39538061 Free PMC article.
-
Molecular mechanisms involved in farnesol-induced apoptosis.Cancer Lett. 2010 Jan 28;287(2):123-35. doi: 10.1016/j.canlet.2009.05.015. Epub 2009 Jun 10. Cancer Lett. 2010. PMID: 19520495 Free PMC article. Review.
-
Safety and efficacy of α,β-unsaturated straight-chain and branched-chain aliphatic primary alcohols, aldehydes, acids and esters belonging to chemical group 3 when used as flavourings for all animal species.EFSA J. 2016 Jun 24;14(6):e04512. doi: 10.2903/j.efsa.2016.4512. eCollection 2016 Jun. EFSA J. 2016. PMID: 40007828 Free PMC article.
-
Importance and Relevance of Phytochemicals Present in Galenia africana.Scientifica (Cairo). 2022 Jan 25;2022:5793436. doi: 10.1155/2022/5793436. eCollection 2022. Scientifica (Cairo). 2022. PMID: 35186343 Free PMC article. Review.
References
-
- Grunler J., Ericsson J., Dallner G. Branch-point reactions in the biosynthesis of cholesterol, dolichol, ubiquinone and prenylated proteins. Biochim. Biophys. Acta. 1994;1212:259–277. - PubMed
-
- Parmryd I., Dallner G. Organization of isoprenoid biosynthesis. Biochem. Soc. Trans. 1996;24:677–682. - PubMed
-
- Wargovich M. J., Jimenez A., McKee K., Steele V. E., Velasco M., Woods J., Price R., Gray K., Kelloff G. J. Efficacy of potential chemopreventive agents on rat colon aberrant crypt formation and progression. Carcinogenesis. 2000;21:1149–1155. - PubMed
-
- Vigushin D. M., Poon G. K., Boddy A., English J., Halbert G. W., Pagonis C., Jarman M., Coombes R. C. Phase I and pharmacokinetic study of D-limonene in patients with advanced cancer. Cancer Chemother. Pharmacol. 1998;42:111–117. - PubMed
-
- Poon G. K., Vigushin D., Griggs L. J., Rowlands M. G., Coombes R. C., Jarman M. Identification and characterization of limonene metabolites in patients with advanced cancer by liquid chromatography mass spectrometry. Drug Metab. Dispos. 1996;24:565–571. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

