Superiority of thyroid peroxidase DNA over protein immunization in replicating human thyroid autoimmunity in HLA-DRB1*0301 (DR3) transgenic mice
- PMID: 15320899
- PMCID: PMC1809151
- DOI: 10.1111/j.1365-2249.2004.02553.x
Superiority of thyroid peroxidase DNA over protein immunization in replicating human thyroid autoimmunity in HLA-DRB1*0301 (DR3) transgenic mice
Abstract
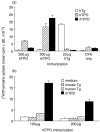
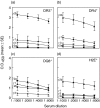
Murine experimental autoimmune thyroiditis (EAT), characterized by thyroid destruction after immunization with thyroglobulin (Tg), has long been a useful model of organ-specific autoimmune disease. More recently, porcine thyroid peroxidase (pTPO) has also been shown to induce thyroiditis, but these results have not been confirmed. When (C57BL/6 x CBA)F(1) mice, recently shown to be susceptible to mouse TPO-induced EAT, were immunized with plasmid DNA to human TPO (hTPO) and cytokines IL-12 or GM-CSF, significant antibody (Ab) titres were generated, but minimal thyroiditis was detected in one mouse only from the TPO + GM-CSF immunized group. However, after TPO DNA immunization of HLA-DR3 transgenic class II-deficient NOD mice, thyroiditis was present in 23% of mice injected with TPO + IL-12 or GM-CSF. We also used another marker for assessing the closeness of the model to human thyroid autoimmunity by examining the epitope profile of the anti-TPO Abs to immunodominant determinants on TPO. Remarkably, the majority of the anti-TPO Abs was directed to immunodominant regions A and B, demonstrating the close replication of the model to human autoimmunity. TPO protein immunizations of HLA-DR3 transgenic mice with recombinant hTPO did not result in thyroiditis, nor did immunization of other mice expressing HLA class II transgenes HLA-DR4 or HLA-DQ8, with differential susceptibility to Tg-induced EAT. Moreover, our efforts to duplicate exactly the experimental procedures used with pTPO also failed to induce thyroiditis. The success of hTPO plasmid DNA immunization of DR3(+) mice, similar to our reports on Tg-induced thyroiditis and thyrotropin receptor DNA-induced Graves' hyperthyroidism, underscores the importance of DR3 genes for all three major thyroid antigens, and provides another humanized model to study autoimmune thyroid disease.
Figures



Similar articles
-
Coexpression of susceptible and resistant HLA class II transgenes in murine experimental autoimmune thyroiditis: DQ8 molecules downregulate DR3-mediated thyroiditis.J Autoimmun. 2002 May;18(3):213-20. doi: 10.1006/jaut.2002.0587. J Autoimmun. 2002. PMID: 12126634
-
Depletion of CD4+CD25+ regulatory T cells exacerbates sodium iodide-induced experimental autoimmune thyroiditis in human leucocyte antigen DR3 (DRB1*0301) transgenic class II-knock-out non-obese diabetic mice.Clin Exp Immunol. 2007 Mar;147(3):547-54. doi: 10.1111/j.1365-2249.2006.03303.x. Clin Exp Immunol. 2007. PMID: 17302906 Free PMC article.
-
Application of HLA class II transgenic mice to study autoimmune regulation.Thyroid. 2007 Oct;17(10):995-1003. doi: 10.1089/thy.2007.0196. Thyroid. 2007. PMID: 17900224 Review.
-
Flexibility of TCR repertoire and permissiveness of HLA-DR3 molecules in experimental autoimmune thyroiditis in nonobese diabetic mice.J Autoimmun. 2001 Aug;17(1):7-15. doi: 10.1006/jaut.2001.0528. J Autoimmun. 2001. PMID: 11488633
-
HLA and H2 class II transgenic mouse models to study susceptibility and protection in autoimmune thyroid disease.Autoimmunity. 2003 Sep-Nov;36(6-7):397-404. doi: 10.1080/08916930310001603028. Autoimmunity. 2003. PMID: 14669947 Review.
Cited by
-
Antibodies to thyroid peroxidase arise spontaneously with age in NOD.H-2h4 mice and appear after thyroglobulin antibodies.Endocrinology. 2010 Sep;151(9):4583-93. doi: 10.1210/en.2010-0321. Epub 2010 Jun 23. Endocrinology. 2010. PMID: 20573721 Free PMC article.
-
cDNA immunization of mice with human thyroglobulin generates both humoral and T cell responses: a novel model of thyroid autoimmunity.PLoS One. 2011 Apr 29;6(4):e19200. doi: 10.1371/journal.pone.0019200. PLoS One. 2011. PMID: 21559421 Free PMC article.
-
Breaking tolerance to thyroid antigens: changing concepts in thyroid autoimmunity.Endocr Rev. 2014 Feb;35(1):59-105. doi: 10.1210/er.2013-1055. Epub 2013 Dec 4. Endocr Rev. 2014. PMID: 24091783 Free PMC article. Review.
-
New approaches for predicting T cell-mediated drug reactions: A role for inducible and potentially preventable autoimmunity.J Allergy Clin Immunol. 2015 Aug;136(2):252-7. doi: 10.1016/j.jaci.2015.06.024. J Allergy Clin Immunol. 2015. PMID: 26254052 Free PMC article. Review.
References
-
- Jacobson DL, Gange SJ, Rose NR, Graham NMH. Epidemiology and estimated population burden of selected autoimmune diseases in the United States. Clin Immunol Immunopathol. 1997;84:223–43. - PubMed
-
- Weetman AP, McGregor AM. Autoimmune thyroid disease: further developments in our understanding. Endocr Rev. 1994;15:788–830. - PubMed
-
- Banga JP. Developments in our understanding of the structure of thyroid peroxidase and the relevance of these findings to autoimmunity. Curr Opin Endocrinol Diabetes. 1998;5:275–81.
-
- Ruf J, Toubert M-E, Czarnocka B, Durand-Gorde J-M, Ferrand M, Carayon P. Relationship between immunological structure and biochemical properties of human thyroid peroxidase. Endocrinology. 1989;125:1211–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

