Effects of Clostridium perfringens alpha-toxin (PLC) and perfringolysin O (PFO) on cytotoxicity to macrophages, on escape from the phagosomes of macrophages, and on persistence of C. perfringens in host tissues
- PMID: 15322015
- PMCID: PMC517428
- DOI: 10.1128/IAI.72.9.5204-5215.2004
Effects of Clostridium perfringens alpha-toxin (PLC) and perfringolysin O (PFO) on cytotoxicity to macrophages, on escape from the phagosomes of macrophages, and on persistence of C. perfringens in host tissues
Abstract
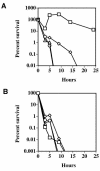
Clostridium perfringens is the most common cause of clostridial myonecrosis (gas gangrene). Polymorphonuclear cells (PMNs) appear to play only a minor role in preventing the onset of myonecrosis in a mouse animal model of the disease (unpublished results). However, the importance of macrophages in the host defense against C. perfringens infections is still unknown. Two membrane-active toxins produced by the anaerobic C. perfringens, alpha-toxin (PLC) and perfringolysin O (PFO), are thought to be important in the pathogenesis of gas gangrene and the lack of phagocytic cells at the site of infection. Therefore, C. perfringens mutants lacking PFO and PLC were examined for their relative cytotoxic effects on macrophages, their ability to escape the phagosome of macrophages, and their persistence in mouse tissues. C. perfringens survival in the presence of mouse peritoneal macrophages was dependent on both PFO and PLC. PFO was shown to be the primary mediator of C. perfringens-dependent cytotoxicity to macrophages. Escape of C. perfringens cells from phagosomes of macrophage-like J774-33 cells and mouse peritoneal macrophages was mediated by either PFO or PLC, although PFO seemed to play a more important role in escape from the phagosome in peritoneal macrophages. At lethal doses (10(9)) of bacteria only PLC was necessary for the onset of myonecrosis, while at sublethal doses (10(6)) both PFO and PLC were necessary for survival of C. perfringens in mouse muscle tissue. These results suggest PFO-mediated cytotoxicity toward macrophages and the ability to escape macrophage phagosomes may be important factors in the ability of C. perfringens to survive in host tissues when bacterial numbers are low relative to those of phagocytic cells, e.g., early in an infection.
Figures

Similar articles
-
Synergistic effects of alpha-toxin and perfringolysin O in Clostridium perfringens-mediated gas gangrene.Infect Immun. 2001 Dec;69(12):7904-10. doi: 10.1128/IAI.69.12.7904-7910.2001. Infect Immun. 2001. PMID: 11705975 Free PMC article.
-
Cross-complementation of Clostridium perfringens PLC and Clostridium septicum alpha-toxin mutants reveals PLC is sufficient to mediate gas gangrene.Microbes Infect. 2009 Mar;11(3):413-8. doi: 10.1016/j.micinf.2009.01.002. Epub 2009 Jan 20. Microbes Infect. 2009. PMID: 19284973
-
Sugar inhibits the production of the toxins that trigger clostridial gas gangrene.Microb Pathog. 2012 Jan;52(1):85-91. doi: 10.1016/j.micpath.2011.10.008. Epub 2011 Nov 4. Microb Pathog. 2012. PMID: 22079896
-
Role of Clostridium perfringens phospholipase C in the pathogenesis of gas gangrene.Toxicon. 2003 Dec 15;42(8):979-86. doi: 10.1016/j.toxicon.2003.11.013. Toxicon. 2003. PMID: 15019495 Review.
-
Perfringolysin O: The Underrated Clostridium perfringens Toxin?Toxins (Basel). 2015 May 14;7(5):1702-21. doi: 10.3390/toxins7051702. Toxins (Basel). 2015. PMID: 26008232 Free PMC article. Review.
Cited by
-
Decreasing Transmembrane Segment Length Greatly Decreases Perfringolysin O Pore Size.J Membr Biol. 2015 Jun;248(3):517-27. doi: 10.1007/s00232-015-9798-5. Epub 2015 Apr 8. J Membr Biol. 2015. PMID: 25850715
-
Engineered nanoparticles mimicking cell membranes for toxin neutralization.Adv Drug Deliv Rev. 2015 Aug 1;90:69-80. doi: 10.1016/j.addr.2015.04.001. Epub 2015 Apr 11. Adv Drug Deliv Rev. 2015. PMID: 25868452 Free PMC article. Review.
-
Arcanobacterium haemolyticum Phospholipase D Enzymatic Activity Promotes the Hemolytic Activity of the Cholesterol-Dependent Cytolysin Arcanolysin.Toxins (Basel). 2018 May 23;10(6):213. doi: 10.3390/toxins10060213. Toxins (Basel). 2018. PMID: 29882842 Free PMC article.
-
Pathogenicity and virulence of Clostridium perfringens.Virulence. 2021 Dec;12(1):723-753. doi: 10.1080/21505594.2021.1886777. Virulence. 2021. PMID: 33843463 Free PMC article.
-
Alpha-toxin of Clostridium perfringens is not an essential virulence factor in necrotic enteritis in chickens.Infect Immun. 2006 Nov;74(11):6496-500. doi: 10.1128/IAI.00806-06. Epub 2006 Aug 21. Infect Immun. 2006. PMID: 16923791 Free PMC article.
References
-
- Aderem, A., and D. M. Underhill. 1999. Mechanisms of phagocytosis in macrophages. Annu. Rev. Immunol. 17:593-623. - PubMed
-
- Awad, M. M., A. E. Bryant, D. L. Stevens, and J. I. Rood. 1995. Virulence studies on chromosomal alpha-toxin and theta-toxin mutants constructed by allelic exchange provide genetic evidence for the essential role of alpha-toxin in Clostridium perfringens-mediated gas gangrene. Mol. Microbiol. 15:191-202. - PubMed
-
- Bryant, A. E., R. Bergstrom, G. A. Zimmerman, J. L. Salyer, H. R. Hill, R. K. Tweten, H. Sato, and D. L. Stevens. 1993. Clostridium perfringens invasiveness is enhanced by effects of theta toxin upon PMNL structure and function: the roles of leukocytotoxicity and expression of CD11/CD18 adherence glycoprotein. FEMS Immunol. Med. Microbiol. 7:321-336. - PubMed
-
- Bryant, A. E., R. Y. Chen, Y. Nagata, Y. Wang, C. H. Lee, S. Finegold, P. H. Guth, and D. L. Stevens. 2000. Clostridial gas gangrene. I. Cellular and molecular mechanisms of microvascular dysfunction induced by exotoxins of Clostridium perfringens. J. Infect. Dis. 182:799-807. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

