Tau induces cooperative Taxol binding to microtubules
- PMID: 15326286
- PMCID: PMC516494
- DOI: 10.1073/pnas.0402928101
Tau induces cooperative Taxol binding to microtubules
Abstract
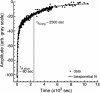
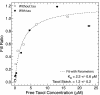
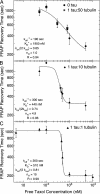
Taxol and tau are two ligands that stabilize the microtubule (MT) lattice. Taxol is an anti-mitotic drug that binds beta tubulin in the MT interior. Tau is a MT-associated protein that binds both alpha and beta tubulin on the MT exterior. Both Taxol and tau reduce MT dynamics and promote tubulin polymerization. Tau alone also acts to bundle, stiffen, and space MTs. A structural study recently suggested that Taxol and tau may interact by binding to the same site. Using fluorescence recovery after photobleaching, we find that tau induces Taxol to bind MTs cooperatively depending on the tau concentration. We develop a model that correctly fits the data in the absence of tau, yields the equilibrium dissociation constant of approximately 2 microM, and determines the escape rate of Taxol through one pore to be 1.7 x 10(3) (M x s)(-1). Extension of the model yields a measure of Taxol cooperativity with a Hill coefficient of at least 15 when tau is present at a 1:1 molar ratio with tubulin.
Copyright 2004 The National Academy of Sciencs of the USA
Figures

Similar articles
-
Tau-isoform dependent enhancement of taxol mobility through microtubules.Arch Biochem Biophys. 2008 Oct 1;478(1):119-26. doi: 10.1016/j.abb.2008.07.020. Epub 2008 Jul 26. Arch Biochem Biophys. 2008. PMID: 18691553
-
Molecular mechanisms of Tau binding to microtubules and its role in microtubule dynamics in live cells.J Cell Sci. 2013 Jul 1;126(Pt 13):2810-9. doi: 10.1242/jcs.120832. Epub 2013 May 9. J Cell Sci. 2013. PMID: 23659998
-
Repeat motifs of tau bind to the insides of microtubules in the absence of taxol.EMBO J. 2003 Jan 2;22(1):70-7. doi: 10.1093/emboj/cdg001. EMBO J. 2003. PMID: 12505985 Free PMC article.
-
Minireview - Microtubules and Tubulin Oligomers: Shape Transitions and Assembly by Intrinsically Disordered Protein Tau and Cationic Biomolecules.Langmuir. 2019 Dec 3;35(48):15970-15978. doi: 10.1021/acs.langmuir.9b02208. Epub 2019 Oct 2. Langmuir. 2019. PMID: 31539262 Free PMC article. Review.
-
FRET and FRAP imaging: approaches to characterise tau and stathmin interactions with microtubules in cells.Biol Cell. 2013 Apr;105(4):149-61. doi: 10.1111/boc.201200060. Epub 2013 Feb 15. Biol Cell. 2013. PMID: 23312015 Review.
Cited by
-
Microtubule-associated protein tau: a marker of paclitaxel sensitivity in breast cancer.Proc Natl Acad Sci U S A. 2005 Jun 7;102(23):8315-20. doi: 10.1073/pnas.0408974102. Epub 2005 May 24. Proc Natl Acad Sci U S A. 2005. PMID: 15914550 Free PMC article.
-
Evaluation of prognostic and predictive value of microtubule associated protein tau in two independent cohorts.Breast Cancer Res. 2011 Nov 2;13(5):R85. doi: 10.1186/bcr2937. Breast Cancer Res. 2011. PMID: 21888627 Free PMC article.
-
The Distance between N and C Termini of Tau and of FTDP-17 Mutants Is Modulated by Microtubule Interactions in Living Cells.Front Mol Neurosci. 2017 Jun 30;10:210. doi: 10.3389/fnmol.2017.00210. eCollection 2017. Front Mol Neurosci. 2017. PMID: 28713242 Free PMC article.
-
The zinc dyshomeostasis hypothesis of Alzheimer's disease.PLoS One. 2012;7(3):e33552. doi: 10.1371/journal.pone.0033552. Epub 2012 Mar 23. PLoS One. 2012. PMID: 22457776 Free PMC article.
-
Microtubule-Binding Proteins as Promising Biomarkers of Paclitaxel Sensitivity in Cancer Chemotherapy.Med Res Rev. 2016 Mar;36(2):300-12. doi: 10.1002/med.21378. Epub 2015 Sep 1. Med Res Rev. 2016. PMID: 26332739 Free PMC article. Review.
References
-
- Jordan, M. A. (2002) Curr. Med. Chem. Anti-Cancer Agents 2, 1–17. - PubMed
-
- Abal, M., Andreau, J. M. & Barasoain, I. (2003) Curr. Cancer Drug Targets 3, 193–203. - PubMed
-
- Schiff, P. B., Fant, J. & Horwitz, S. B. (1979) Nature 277, 665–667. - PubMed
-
- Kumar, N. (1981) J. Biol. Chem. 256, 10435–10441. - PubMed
-
- Howard, W. D. & Timasheff, S. N. (1988) J. Biol. Chem. 263, 1342–1346. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

