Characterization of human cytochrome P450 enzymes catalyzing domperidone N-dealkylation and hydroxylation in vitro
- PMID: 15327587
- PMCID: PMC1884565
- DOI: 10.1111/j.1365-2125.2004.02156.x
Characterization of human cytochrome P450 enzymes catalyzing domperidone N-dealkylation and hydroxylation in vitro
Abstract
Aims: To confirm the identity of the major metabolites of domperidone and to characterize the cytochrome P450s (CYPs) involved in their formation.
Methods: Human liver microsomes (HLMs) were used to characterize the kinetics of domperidone metabolism and liquid chromatography-mass spectrometry to identify the products. Isoform-specific chemical inhibitors, correlation analysis and expressed human CYP genes were used to identify the CYPs involved in domperidone oxidation.
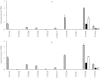
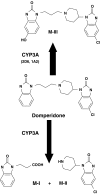
Results: In HLMs, domperidone underwent hydroxylation to form 5-hydroxydomperidone (MIII) and N-dealkylation to form 2,3-dihydro-2-oxo-1H-benzimidazole-1-propionic acid (MI) and 5-chloro-4-piperidinyl-1,3-dihydro-benzimidazol-2-one (MII). The formation of all three metabolites (n = 4 HLMs) followed apparent Michaelis-Menten kinetics. The mean Km values for MI, MII and MIII formation were 12.4, 11.9, and 12.6 micro m, respectively. In a panel of HLMs (n = 10), the rate of domperidone (5 microm and 50 microm) metabolism correlated with the activity of CYP3A (r > 0.94; P < 0.0001). Only ketoconazole (1 microm) (by 87%) and troleandomycin (50 microm) (by 64%) inhibited domperidone (5 microm) metabolism in HLMs. Domperidone (5 and 50 microm) hydroxylation and N-dealkylation was catalyzed by expressed CYP3A4 at a higher rate than the other CYPs. CYP1A2, 2B6, 2C8 and 2D6 also hydroxylated domperidone
Conclusions: CYP3A-catalyzed N-dealkylation and aromatic hydroxylation are the major routes for domperidone metabolism. The drug would be expected to demonstrate highly variable bioavailability due to hepatic, and possibly intestinal first-pass metabolism after oral administration. Increased risk of adverse effects might be anticipated during concomitant administration with CYP3A inhibitors, as well as decreased efficacy with inducers of this enzyme.
Figures

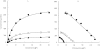
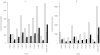
 ), MIII (□)
), MIII (□)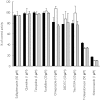
 )
)
 )
)
 ), MII (▪), MI (□)
), MII (▪), MI (□)
References
-
- Brogden RN, Carmine AA, Heel RC, Speight TM, Avery GS. Domperidone. A review of its pharmacological activity, pharmacokinetics and therapeutic efficacy in the symptomatic treatment of chronic dyspepsia and as an antiemetic. Drugs. 1982;24:360–400. - PubMed
-
- Prakash A, Wagstaff AJ. Domperidone. A review of its use in diabetic gastropathy. Drugs. 1998;56:429–45. - PubMed
-
- Barone JA. Domperidone: a peripherally acting dopamine2-receptor antagonist. Ann Pharmacother. 1999;33:429–40. - PubMed
-
- Woosley RL, Chen Y, Freiman JP, Gillis RA. Mechanism of the cardiotoxic actions of terfenadine. JAMA. 1993;269:1532–6. - PubMed
-
- Cubeddu LX. QT prolongation and fatal arrhythmias. a review of clinical implications and effects of drugs. Am J Ther. 2003;10:452–7. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

