Clinical emergence of entecavir-resistant hepatitis B virus requires additional substitutions in virus already resistant to Lamivudine
- PMID: 15328117
- PMCID: PMC514758
- DOI: 10.1128/AAC.48.9.3498-3507.2004
Clinical emergence of entecavir-resistant hepatitis B virus requires additional substitutions in virus already resistant to Lamivudine
Abstract
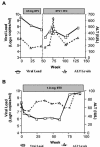
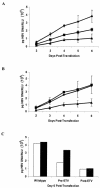
Entecavir (ETV) exhibits potent antiviral activity in patients chronically infected with wild-type or lamivudine (3TC)-resistant (3TC(r)) hepatitis B virus (HBV). Among the patients treated in phase II ETV clinical trials, two patients for whom previous therapies had failed exhibited virologic breakthrough while on ETV. Isolates from these patients (arbitrarily designated patients A and B) were analyzed genotypically for emergent substitutions in HBV reverse transcriptase (RT) and phenotypically for reduced susceptibility in cultures and in HBV polymerase assays. After 54 weeks of 3TC therapy, patient A (AI463901-A) received 0.5 mg of ETV for 52 weeks followed by a combination of ETV and 100 mg of 3TC for 89 weeks. Viral rebound occurred at 133 weeks after ETV was started. The 3TC(r) RT substitutions rtV173L, rtL180M, and rtM204V were present at study entry, and the additional substitutions rtI169T and rtM250V emerged during ETV-3TC combination treatment. Reduced ETV susceptibility in vitro required the rtM250V substitution in addition to the 3TC(r) substitutions. For liver transplant patient B (AI463015-B), previous famciclovir, ganciclovir, foscarnet, and 3TC therapies had failed, and RT changes rtS78S/T, rtV173L, rtL180M, rtT184S, and rtM204V were present at study entry. Viral rebound occurred after 76 weeks of therapy with ETV at 1.0 mg, with the emergence of rtT184G, rtI169T, and rtS202I substitutions within the preexisting 3TC(r) background. Reduced susceptibility in vitro was highest when both the rtT184G and the rtS202I changes were combined with the 3TC(r) substitutions. In summary, infrequent ETV resistance can emerge during prolonged therapy, with selection of additional RT substitutions within a 3TC(r) HBV background, leading to reduced ETV susceptibility and treatment failure.
Figures
Similar articles
-
Two-year assessment of entecavir resistance in Lamivudine-refractory hepatitis B virus patients reveals different clinical outcomes depending on the resistance substitutions present.Antimicrob Agents Chemother. 2007 Mar;51(3):902-11. doi: 10.1128/AAC.00833-06. Epub 2006 Dec 18. Antimicrob Agents Chemother. 2007. PMID: 17178796 Free PMC article. Clinical Trial.
-
Efficacies of entecavir against lamivudine-resistant hepatitis B virus replication and recombinant polymerases in vitro.Antimicrob Agents Chemother. 2002 Aug;46(8):2525-32. doi: 10.1128/AAC.46.8.2525-2532.2002. Antimicrob Agents Chemother. 2002. PMID: 12121928 Free PMC article.
-
Impact of hepatitis B e antigen-suppressing mutations on the replication efficiency of entecavir-resistant hepatitis B virus strains.J Viral Hepat. 2011 Nov;18(11):804-14. doi: 10.1111/j.1365-2893.2010.01378.x. Epub 2010 Sep 30. J Viral Hepat. 2011. PMID: 20887378
-
Effects of entecavir and lamivudine for hepatitis B decompensated cirrhosis: meta-analysis.World J Gastroenterol. 2013 Oct 21;19(39):6665-78. doi: 10.3748/wjg.v19.i39.6665. World J Gastroenterol. 2013. PMID: 24151397 Free PMC article. Review.
-
Naturally occurring hepatitis B virus reverse transcriptase mutations related to potential antiviral drug resistance and liver disease progression.World J Gastroenterol. 2018 Apr 28;24(16):1708-1724. doi: 10.3748/wjg.v24.i16.1708. World J Gastroenterol. 2018. PMID: 29713126 Free PMC article. Review.
Cited by
-
Two cases of development of entecavir resistance during entecavir treatment for nucleoside-naive chronic hepatitis B.Hepatol Int. 2009 Jun;3(2):403-10. doi: 10.1007/s12072-008-9108-8. Epub 2008 Dec 9. Hepatol Int. 2009. PMID: 19669367 Free PMC article.
-
Novel lamivudine resistance.Antimicrob Agents Chemother. 2007 Dec;51(12):4533, author reply 4533-4. doi: 10.1128/AAC.00840-07. Antimicrob Agents Chemother. 2007. PMID: 18025122 Free PMC article. No abstract available.
-
Options for the management of antiviral resistance during hepatitis B therapy: reflections on battles over a decade.Clin Mol Hepatol. 2013 Sep;19(3):195-209. doi: 10.3350/cmh.2013.19.3.195. Epub 2013 Sep 30. Clin Mol Hepatol. 2013. PMID: 24133659 Free PMC article. Review.
-
Resistance mutations of hepatitis B virus in entecavir-refractory patients.Hepatol Commun. 2017 Mar 9;1(2):110-121. doi: 10.1002/hep4.1022. eCollection 2017 Apr. Hepatol Commun. 2017. PMID: 29404449 Free PMC article.
-
The Clinical Value of Oxymatrine in Preventing Lamivudine Induced YMDD Mutation: A Meta-Analysis.Evid Based Complement Alternat Med. 2015;2015:971616. doi: 10.1155/2015/971616. Epub 2015 Oct 5. Evid Based Complement Alternat Med. 2015. PMID: 26508988 Free PMC article. Review.
References
-
- Allen, M. I., M. Deslauriers, C. W. Andrews, G. A. Tipples, K. A. Walters, D. L. Tyrrell, N. Brown, L. D. Condreay, et al. 1998. Identification and characterization of mutations in hepatitis B virus resistant to lamivudine. Hepatology 27:1670-1677. - PubMed
-
- Angus, P., R. Vaughan, S. Xiong, H. Yang, W. Delaney, C. Gibbs, C. Brosgart, D. Colledge, R. Edwards, A. Ayres, A. Bartholomeusz, and S. Locarnini. 2003. Resistance to adefovir dipivoxil therapy associated with the selection of a novel mutation in the HBV polymerase. Gastroenterology 125:292-297. - PubMed
-
- Aye, T. T., A. Bartholomeusz, T. Shaw, S. Bowden, A. Breschkin, J. McMillan, P. Angus, and S. Locarnini. 1997. Hepatitis B virus polymerase mutations during antiviral therapy in a patient following liver transplantation. J. Hepatol. 26:1148-1153. - PubMed
-
- Chang, T.-T., S. Hadziyannis, J. Cianciara, M. Rizzeto, E. Schiff, G. Pastore, K. Klesczewski, G. Denisky, D. DeHertogh, and R. Hindes. 2002. Sustained viral load and ALT reduction following 48 weeks of entecavir treatment in subjects with chronic hepatitis B who have failed lamivudine. Hepatology 36:300A.
-
- Chayama, K., Y. Suzuki, M. Kobayashi, A. Tsubota, M. Hashimoto, Y. Miyano, H. Koike, I. Koida, Y. Arase, S. Saitoh, N. Murashima, K. Ikeda, and H. Kumada. 1998. Emergence and takeover of YMDD motif mutant hepatitis B virus during long-term lamivudine therapy and re-takeover by wild type after cessation of therapy. Hepatology 27:1711-1716. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

