Substance P-mediated modulation of pacemaker properties in the mammalian respiratory network
- PMID: 15329402
- PMCID: PMC6729648
- DOI: 10.1523/JNEUROSCI.1871-04.2004
Substance P-mediated modulation of pacemaker properties in the mammalian respiratory network
Abstract
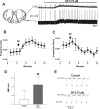
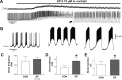
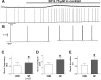
Neuromodulators are integral parts of a neuronal network, and unraveling how these substances alter neuronal activity is critical for understanding how networks generate patterned activity and, ultimately, behavior. In this study, we examined the cellular mechanisms underlying the excitatory action of substance P (SP) on the respiratory network isolated in spontaneously active transverse slice preparation of mice. SP produced a slow depolarization in all recorded inspiratory pacemaker and non-pacemaker neurons. Ion exchange experiments and blockers for different ion channels suggest that the slow depolarization is caused by the activation of a low-threshold TTX-insensitive cationic current that carries mostly Na+. The SP-induced slow depolarization increased tonic discharge in non-pacemaker neurons and primarily enhanced the frequency of bursting in Cd2+-insensitive pacemaker neurons. In the Cd2+-sensitive pacemaker neuron, the burst frequency was not significantly affected, whereas burst duration and amplitude were more enhanced than in Cd2+-insensitive pacemaker neurons. In a subset of non-pacemaker neurons that produced NMDA-dependent subthreshold oscillations, SP caused the production of bursts of action potentials. We conclude that the degree of pacemaker activity in the respiratory network is not fixed but dynamically regulated by neuromodulators such as SP. This finding may have clinical implications for Rett syndrome in which SP levels along with other neuromodulators are decreased in the brainstem.
Figures




Similar articles
-
Identification of two types of inspiratory pacemaker neurons in the isolated respiratory neural network of mice.J Neurophysiol. 2001 Jul;86(1):104-12. doi: 10.1152/jn.2001.86.1.104. J Neurophysiol. 2001. PMID: 11431492
-
Persistent sodium current, membrane properties and bursting behavior of pre-bötzinger complex inspiratory neurons in vitro.J Neurophysiol. 2002 Nov;88(5):2242-50. doi: 10.1152/jn.00081.2002. J Neurophysiol. 2002. PMID: 12424266
-
Endogenous activation of serotonin-2A receptors is required for respiratory rhythm generation in vitro.J Neurosci. 2002 Dec 15;22(24):11055-64. doi: 10.1523/JNEUROSCI.22-24-11055.2002. J Neurosci. 2002. PMID: 12486201 Free PMC article.
-
Determinants of inspiratory activity.Respir Physiol Neurobiol. 2005 Jul 28;147(2-3):145-57. doi: 10.1016/j.resp.2005.05.003. Respir Physiol Neurobiol. 2005. PMID: 15964786 Review.
-
Pacemaker neurons and neuronal networks: an integrative view.Curr Opin Neurobiol. 2004 Dec;14(6):665-74. doi: 10.1016/j.conb.2004.10.011. Curr Opin Neurobiol. 2004. PMID: 15582367 Review.
Cited by
-
Phosphatidylinositol 4,5-bisphosphate regulates inspiratory burst activity in the neonatal mouse preBötzinger complex.J Physiol. 2007 Aug 1;582(Pt 3):1047-58. doi: 10.1113/jphysiol.2007.134577. Epub 2007 Jun 28. J Physiol. 2007. PMID: 17599963 Free PMC article.
-
Respiratory rhythm generation, hypoxia, and oxidative stress-Implications for development.Respir Physiol Neurobiol. 2019 Dec;270:103259. doi: 10.1016/j.resp.2019.103259. Epub 2019 Jul 29. Respir Physiol Neurobiol. 2019. PMID: 31369874 Free PMC article. Review.
-
The hypoxic respiratory response of the pre-Bötzinger complex.Heliyon. 2024 Jul 11;10(14):e34491. doi: 10.1016/j.heliyon.2024.e34491. eCollection 2024 Jul 30. Heliyon. 2024. PMID: 39114066 Free PMC article. Review.
-
Substance P differentially modulates firing rate of solitary complex (SC) neurons from control and chronic hypoxia-adapted adult rats.PLoS One. 2014 Feb 7;9(2):e88161. doi: 10.1371/journal.pone.0088161. eCollection 2014. PLoS One. 2014. PMID: 24516602 Free PMC article.
-
State-dependent interactions between excitatory neuromodulators in the neuronal control of breathing.J Neurosci. 2010 Jun 16;30(24):8251-62. doi: 10.1523/JNEUROSCI.5361-09.2010. J Neurosci. 2010. PMID: 20554877 Free PMC article.
References
-
- Akasu T, Ishimatsu M, Yamada K (1996) Tachykinins cause inward current through NK1 receptors in bullfrog sensory neurons. Brain Res 713: 160-167. - PubMed
-
- Alberi S, Dubois-Dauphin M, Dreifuss JJ, Raggenbass M (1993) Modulation by divalent cations of the current generated by vasopressin in facial motoneurons. Brain Res 624: 326-330. - PubMed
-
- Bell MI, Richardson PJ, Lee K (1998) Characterization of the mechanism of action of tachykinins in rat striatal cholinergic interneurons. Neuroscience 87: 649-658. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
