Repetitive behaviors in monkeys are linked to specific striatal activation patterns
- PMID: 15329403
- PMCID: PMC6729641
- DOI: 10.1523/JNEUROSCI.1072-04.2004
Repetitive behaviors in monkeys are linked to specific striatal activation patterns
Abstract
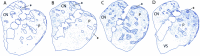
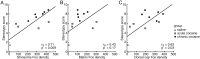
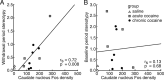
The spontaneous behavior of humans can be altered dramatically by repeated exposure to psychomotor stimulants. We have developed a primate model for analyzing the neurobiology underlying such drug-induced behavioral changes. We performed ethogram-based behavioral assays on squirrel monkeys given single or multiple cocaine treatments, and in the same monkeys made anatomical plots of striatal neurons that were activated to express early-gene proteins. A final cocaine challenge after chronic intermittent exposure to cocaine induced highly patterned behavioral changes in the monkeys, affecting individual behavioral motifs in distinct ways. In the striatum, the challenge dose induced striosome-predominant expression combined with intense dorsal early-gene expression, especially in the putamen. These patterns of gene expression were highly predictive of the levels of stereotypy exhibited by the monkeys in response to cocaine challenge. The total levels of expression, on the other hand, appeared to reflect increased spontaneous behavioral activation during the drug-free period after the cocaine exposure. We suggest that in the primate, compartmentally and regionally specific striatal activation patterns contribute to the striatal modulation of psychostimulant-induced behaviors. These observations in nonhuman primates raise the possibility that monitoring such basal ganglia activity patterns could help to delineate the neural mechanisms underlying drug-induced repetitive behaviors and related syndromes in which stereotypies are manifest.
Figures




Similar articles
-
Striatal patch compartment lesions reduce stereotypy following repeated cocaine administration.Brain Res. 2015 Aug 27;1618:286-98. doi: 10.1016/j.brainres.2015.06.012. Epub 2015 Jun 19. Brain Res. 2015. PMID: 26100338 Free PMC article.
-
Concurrent activation of dopamine D1 and D2 receptors is required to evoke neural and behavioral phenotypes of cocaine sensitization.J Neurosci. 2002 Jul 15;22(14):6218-27. doi: 10.1523/JNEUROSCI.22-14-06218.2002. J Neurosci. 2002. PMID: 12122080 Free PMC article.
-
Network-level changes in expression of inducible Fos-Jun proteins in the striatum during chronic cocaine treatment and withdrawal.Neuron. 1996 Jul;17(1):147-56. doi: 10.1016/s0896-6273(00)80288-3. Neuron. 1996. PMID: 8755486
-
A measure of striatal function predicts motor stereotypy.Nat Neurosci. 2000 Apr;3(4):377-83. doi: 10.1038/73949. Nat Neurosci. 2000. PMID: 10725928
-
Cocaine-induced c-fos messenger RNA is inversely related to dynorphin expression in striatum.J Neurosci. 1993 Dec;13(12):5066-81. doi: 10.1523/JNEUROSCI.13-12-05066.1993. J Neurosci. 1993. PMID: 7504719 Free PMC article.
Cited by
-
Striatal patch compartment lesions reduce stereotypy following repeated cocaine administration.Brain Res. 2015 Aug 27;1618:286-98. doi: 10.1016/j.brainres.2015.06.012. Epub 2015 Jun 19. Brain Res. 2015. PMID: 26100338 Free PMC article.
-
Striatal insights: a cellular and molecular perspective on repetitive behaviors in pathology.Front Cell Neurosci. 2024 Mar 27;18:1386715. doi: 10.3389/fncel.2024.1386715. eCollection 2024. Front Cell Neurosci. 2024. PMID: 38601025 Free PMC article. Review.
-
A novel method for automatic quantification of psychostimulant-evoked route-tracing stereotypy: application to Mus musculus.Psychopharmacology (Berl). 2008 Mar;196(4):591-602. doi: 10.1007/s00213-007-0994-6. Epub 2007 Dec 21. Psychopharmacology (Berl). 2008. PMID: 18097652 Free PMC article.
-
Cortical-striatal integration of cocaine history and prefrontal dysfunction in animal modeling of dual diagnosis.Biol Psychiatry. 2010 Apr 15;67(8):788-92. doi: 10.1016/j.biopsych.2009.09.006. Epub 2009 Oct 31. Biol Psychiatry. 2010. PMID: 19880093 Free PMC article.
-
Cortico-Basal Ganglia reward network: microcircuitry.Neuropsychopharmacology. 2010 Jan;35(1):27-47. doi: 10.1038/npp.2009.93. Neuropsychopharmacology. 2010. PMID: 19675534 Free PMC article. Review.
References
-
- Asin KE, Wirtshafter D, Nikkel A (1996) Amphetamine induces Fos-like immunoreactivity in the striatum of primates. Brain Res 719: 138-142. - PubMed
-
- Baldessarini RJ (1972) Symposium: behavior modification by drugs. I. Pharmacology of the amphetamines. Pediatrics 49: 694-701. - PubMed
-
- Bibb JA, Chen J, Taylor JR, Svenningsson P, Nishi A, Snyder GL, Yan Z, Sagawa ZK, Ouimet CC, Nairn AC, Nestler EJ, Greengard P (2001) Effects of chronic exposure to cocaine are regulated by the neuronal protein Cdk5. Nature 410: 376-380. - PubMed
-
- Breiter HC, Rauch SL, Kwong KK, Baker JR, Weisskoff RM, Kennedy DN, Kendrick AD, Davis TL, Jiang A, Cohen MS, Stern CE, Belliveau JW, Baer L, O'Sullivan RL, Savage CR, Jenike MA, Rosen BR (1996) Functional magnetic resonance imaging of symptom provocation in obsessive-compulsive disorder. Arch Gen Psychiatry 53: 595-606. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R56 NS025529/NS/NINDS NIH HHS/United States
- DA11558/DA/NIDA NIH HHS/United States
- RR00168/RR/NCRR NIH HHS/United States
- DA08037/DA/NIDA NIH HHS/United States
- P51 OD011103/OD/NIH HHS/United States
- R01 DA006303/DA/NIDA NIH HHS/United States
- DA15305/DA/NIDA NIH HHS/United States
- K05 DA015305/DA/NIDA NIH HHS/United States
- R01 DA011558/DA/NIDA NIH HHS/United States
- R01 NS025529/NS/NINDS NIH HHS/United States
- NS25529/NS/NINDS NIH HHS/United States
- R37 DA006303/DA/NIDA NIH HHS/United States
- DA06303/DA/NIDA NIH HHS/United States
- P51 RR000168/RR/NCRR NIH HHS/United States
- K26 RR000168/RR/NCRR NIH HHS/United States
LinkOut - more resources
Full Text Sources
