Arginine mutations within a transmembrane domain of Tar, an Escherichia coli aspartate receptor, can drive homodimer dissociation and heterodimer association in vivo
- PMID: 15330757
- PMCID: PMC1134670
- DOI: 10.1042/BJ20041022
Arginine mutations within a transmembrane domain of Tar, an Escherichia coli aspartate receptor, can drive homodimer dissociation and heterodimer association in vivo
Abstract
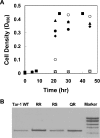
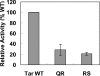
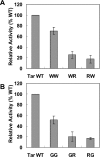
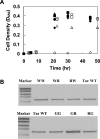
The interactions between the TM (transmembrane) domains of many membrane proteins are important for their proper functioning. Mutations of residues into positively charged ones within TM domains were reported to be involved in many genetic diseases, possibly because these mutations affect the self- and/or hetero-assembly of the corresponding proteins. To our knowledge, despite significant progress in understanding the role of various amino acids in TM-TM interactions in vivo, the direct effect of positively charged residues on these interactions has not been studied. To address this issue, we employed the N-terminal TM domain of the aspartate receptor (Tar-1) as a dimerization model system. We expressed within the ToxR TM assembly system several Tar-1 constructs that dimerize via polar- or non-polar amino acid motifs, and mutated these by replacement with a single arginine residue. Our results have revealed that a mutation in each of the motifs significantly reduced the ability of the TMs to dimerize. Furthermore, a Tar-1 construct that contained two arginine residues was unable to correctly integrate itself into the membrane. Nevertheless, an exogenous synthetic Tar-1 peptide containing these two arginine residues was able to inhibit in vivo the marked dimerization of a mutant Tar-1 construct that contained two glutamate residues at similar positions. This indicates that hetero-assembly of TM domains can be mediated by the interaction of two oppositely charged residues, probably by formation of ion pairs. This study broadens our knowledge regarding the effect of positively charged residues on TM-TM interactions in vivo, and provides a potential therapeutic approach to inhibit uncontrolled dimerization of TM domains caused by mutations of polar amino acids.
Figures
References
-
- Ding P. Z., Wilson T. H. The effect of modifications of the charged residues in the transmembrane helices on the transport activity of the melibiose carrier of Escherichia coli. Biochem. Biophys. Res. Commun. 2001;285:348–354. - PubMed
-
- Burnay M., Crambert G., Kharoubi-Hess S., Geering K., Horisberger J. D. Electrogenicity of Na,K- and H,K-ATPase activity and presence of a positively charged amino acid in the fifth transmembrane segment. J. Biol. Chem. 2003;278:19237–19244. - PubMed
-
- Xu Y., Kakhniashvili D. A., Gremse D. A., Wood D. O., Mayor J. A., Walters D. E., Kaplan R. S. The yeast mitochondrial citrate transport protein. Probing the roles of cysteines, Arg181, and Arg189 in transporter function. J. Biol. Chem. 2000;275:7117–7124. - PubMed
-
- Partridge A. W., Melnyk R. A., Deber C. M. Polar residues in membrane domains of proteins: molecular basis for helix-helix association in a mutant CFTR transmembrane segment. Biochemistry. 2002;41:3647–3653. - PubMed
-
- Partridge A. W., Therien A. G., Deber C. M. Missense mutations in transmembrane domains of proteins: phenotypic propensity of polar residues for human disease. Proteins. 2004;54:648–656. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

