Neuroepithelial oxygen chemoreceptors of the zebrafish gill
- PMID: 15331683
- PMCID: PMC1665292
- DOI: 10.1113/jphysiol.2004.069294
Neuroepithelial oxygen chemoreceptors of the zebrafish gill
Abstract
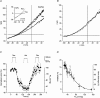
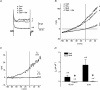
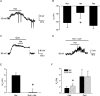
In aquatic vertebrates, hypoxia induces physiological changes that arise principally from O(2) chemoreceptors of the gill. Neuroepithelial cells (NECs) of the zebrafish gill are morphologically similar to mammalian O(2) chemoreceptors (e.g. carotid body), suggesting that they may play a role in initiating the hypoxia response in fish. We describe morphological changes of zebrafish gill NECs following in vivo exposure to chronic hypoxia, and characterize the cellular mechanisms of O(2) sensing in isolated NECs using patch-clamp electrophysiology. Confocal immunofluorescence studies indicated that chronic hypoxia (P(O(2)) = 35 mmHg, 60 days) induced hypertrophy, proliferation and process extension in NECs immunoreactive for serotonin or synaptic vesicle protein (SV2). Under voltage clamp, NECs responded to hypoxia (P(O(2)) = 25-140 mmHg) with a dose-dependent decrease in K(+) current. The current-voltage relationship of the O(2)-sensitive current (I(KO(2))) reversed near E(K) and displayed open rectification. Pharmacological characterization indicated that I(KO(2)) was resistant to 20 mM tetraethylammonium (TEA) and 5 mM 4-aminopyridine (4-AP), but was sensitive to 1 mm quinidine. In current-clamp recordings, hypoxia produced membrane depolarization associated with a conductance decrease; this depolarization was blocked by quinidine, but was insensitive to TEA and 4-AP. These biophysical and pharmacological characteristics suggest that hypoxia sensing in zebrafish gill NECs is mediated by inhibition of a background K(+) conductance, which generates a receptor potential necessary for neurosecretion and activation of sensory pathways in the gill. This appears to be a fundamental mechanism of O(2) sensing that arose early in vertebrate evolution, and was adopted later in mammalian O(2) chemoreceptors.
Figures
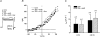
Similar articles
-
Development of oxygen sensing in the gills of zebrafish.J Exp Biol. 2005 Apr;208(Pt 8):1537-49. doi: 10.1242/jeb.01564. J Exp Biol. 2005. PMID: 15802677
-
Serotonergic and cholinergic elements of the hypoxic ventilatory response in developing zebrafish.J Exp Biol. 2013 Mar 1;216(Pt 5):869-80. doi: 10.1242/jeb.079657. Epub 2012 Nov 15. J Exp Biol. 2013. PMID: 23155078
-
Chemoreceptor plasticity and respiratory acclimation in the zebrafish Danio rerio.J Exp Biol. 2006 Apr;209(Pt 7):1261-73. doi: 10.1242/jeb.02058. J Exp Biol. 2006. PMID: 16547298
-
Peripheral chemoreceptors in fish: A brief history and a look ahead.Comp Biochem Physiol A Mol Integr Physiol. 2015 Aug;186:27-38. doi: 10.1016/j.cbpa.2014.09.002. Epub 2014 Sep 16. Comp Biochem Physiol A Mol Integr Physiol. 2015. PMID: 25218943 Review.
-
Neuroepithelial cells of the gill and their role in oxygen sensing.Respir Physiol Neurobiol. 2012 Dec 1;184(3):301-8. doi: 10.1016/j.resp.2012.06.024. Epub 2012 Jul 3. Respir Physiol Neurobiol. 2012. PMID: 22772312 Review.
Cited by
-
Zebrafish (Danio rerio) gill neuroepithelial cells are sensitive chemoreceptors for environmental CO2.J Physiol. 2010 Mar 1;588(Pt 5):861-72. doi: 10.1113/jphysiol.2009.184739. Epub 2010 Jan 5. J Physiol. 2010. PMID: 20051495 Free PMC article.
-
Shaping the cardiac response to hypoxia: NO and its partners in teleost fish.Curr Res Physiol. 2022 Apr 4;5:193-202. doi: 10.1016/j.crphys.2022.03.006. eCollection 2022. Curr Res Physiol. 2022. PMID: 35434651 Free PMC article.
-
AMP-activated protein kinase and the regulation of Ca2+ signalling in O2-sensing cells.J Physiol. 2006 Jul 1;574(Pt 1):113-23. doi: 10.1113/jphysiol.2006.108381. Epub 2006 May 18. J Physiol. 2006. PMID: 16709639 Free PMC article. Review.
-
Fish Behavior as a Neural Proxy to Reveal Physiological States.Front Physiol. 2022 Jul 13;13:937432. doi: 10.3389/fphys.2022.937432. eCollection 2022. Front Physiol. 2022. PMID: 35910555 Free PMC article. Review.
-
Out of thin air: sensory detection of oxygen and carbon dioxide.Neuron. 2011 Jan 27;69(2):194-202. doi: 10.1016/j.neuron.2010.12.018. Neuron. 2011. PMID: 21262460 Free PMC article. Review.
References
-
- Barrionuevo WR, Burggren WW. O2 consumption and heart rate in developing zebrafish (Danio rerio): influence of temperature and ambient O2. Am J Physiol Regul Integr Comp Physiol. 1999;276:R505–R513. - PubMed
-
- Bisgard GE. Carotid body mechanisms in acclimatization to hypoxia. Respir Physiol. 2000;121:237–246. - PubMed
-
- Bradford CS, Sun L, Collodi P, Barnes DW. Cell cultures from zebrafish embryos and adult tissues. Mol Mar Biol Biotechnol. 1994;3:78–86. - PubMed
-
- Brockerhoff SE, Dowling JE, Hurley JB. Zebrafish retinal mutants. Vision Res. 1998;38:1335–1339. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

