Escherichia coli glutamate- and arginine-dependent acid resistance systems increase internal pH and reverse transmembrane potential
- PMID: 15342572
- PMCID: PMC515135
- DOI: 10.1128/JB.186.18.6032-6041.2004
Escherichia coli glutamate- and arginine-dependent acid resistance systems increase internal pH and reverse transmembrane potential
Abstract
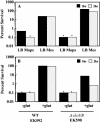
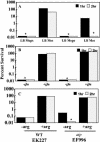
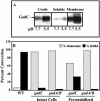
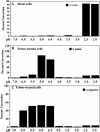
Due to the acidic nature of the stomach, enteric organisms must withstand extreme acid stress for colonization and pathogenesis. Escherichia coli contains several acid resistance systems that protect cells to pH 2. One acid resistance system, acid resistance system 2 (AR2), requires extracellular glutamate, while another (AR3) requires extracellular arginine. Little is known about how these systems protect cells from acid stress. AR2 and AR3 are thought to consume intracellular protons through amino acid decarboxylation. Antiport mechanisms then exchange decarboxylation products for new amino acid substrates. This form of proton consumption could maintain an internal pH (pHi) conducive to cell survival. The model was tested by estimating the pHi and transmembrane potential (DeltaPsi) of cells acid stressed at pH 2.5. During acid challenge, glutamate- and arginine-dependent systems elevated pHi from 3.6 to 4.2 and 4.7, respectively. However, when pHi was manipulated to 4.0 in the presence or absence of glutamate, only cultures challenged in the presence of glutamate survived, indicating that a physiological parameter aside from pHi was also important. Measurements of DeltaPsi indicated that amino acid-dependent acid resistance systems help convert membrane potential from an inside negative to inside positive charge, an established acidophile strategy used to survive extreme acidic environments. Thus, reversing DeltaPsi may be a more important acid resistance strategy than maintaining a specific pHi value.
Figures


References
-
- Accardi, A., and C. Miller. 2004. Secondary active transport mediated by a prokaryotic homologue of ClC Cl− channels. Nature 427:803-807. - PubMed
-
- Anantharam, V., M. J. Allison, and P. C. Maloney. 1989. Oxalate:formate exchange. The basis for energy coupling in Oxalobacter. J. Biol. Chem. 264:7244-7250. - PubMed
-
- Applebaum, D., D. L. Sabo, E. H. Fischer, and D. R. Morris. 1975. Biodegradative ornithine decarboxylase of Escherichia coli. Purification, properties, and pyridoxal 5′-phosphate binding site. Biochemistry 14:3675-3681. - PubMed
-
- Atkinson, W. H., and H. H. Winkler. 1981. A centrifugal filtration method for the study of transport of nicotinamide adenine and pyruvate by Rickettsia prowazekii, p. 411-420. In W. Burgerdorfer and R. L. Anacker (ed.), Rickettsiae and rickettsial disease. Academic Press, Inc., New York, N.Y.
-
- Boeker, E. A., and E. E. Snell. 1972. Amino acid decarboxylases. Enzymes 6:217-250.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

