The site of primary T cell activation is a determinant of the balance between intrahepatic tolerance and immunity
- PMID: 15343389
- PMCID: PMC514586
- DOI: 10.1172/JCI21593
The site of primary T cell activation is a determinant of the balance between intrahepatic tolerance and immunity
Abstract
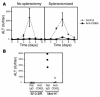
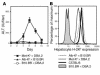
Hepatic immunobiology is paradoxical: although the liver possesses unusual tolerogenic properties, it is also the site of effective immune responses against multiple pathogens and subject to immune-mediated pathology. The mechanisms underlying this dichotomy remain unclear. Following previous work demonstrating that the liver may act as a site of primary T cell activation, we demonstrate here that the balance between immunity and tolerance in this organ is established by competition for primary activation of CD8+ T cells between the liver and secondary lymphoid tissues, with the immune outcome determined by the initial site of activation. Using a transgenic mouse model in which antigen is expressed within both liver and lymph nodes, we show that while naive CD8+ T cells activated within the lymph nodes were capable of mediating hepatitis, cells undergoing primary activation within the liver exhibited defective cytotoxic function and shortened half-life and did not mediate hepatocellular injury. The implications of these novel findings may pertain not only to the normal maintenance of peripheral tolerance, but also to hepatic allograft tolerance and the immunopathogenesis of chronic viral hepatitis.
Figures




References
-
- Bertolino P, Glimpel G, Lemon SM. Hepatic inflammation and immunity: a summary of a conference on the function of the immune system within the liver. Hepatology. 2000;31:1374–1378. - PubMed
-
- Crispe IN. Hepatic T cells and liver tolerance. Nat. Rev. Immunol. 2003;3:51–62. - PubMed
-
- Calne RY, et al. Induction of immunological tolerance by porcine liver allografts. Nature. 1969;223:472–476. - PubMed
-
- Sun J, McCaughan GW, Gallagher ND, Sheil AG, Bishop GA. Deletion of spontaneous rat liver allograft acceptance by donor irradiation. Transplantation. 1995;60:233–236. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

