Cyclo-oxygenase type 2-dependent prostaglandin E2 secretion is involved in retrovirus-induced T-cell dysfunction in mice
- PMID: 15344910
- PMCID: PMC1134132
- DOI: 10.1042/BJ20031859
Cyclo-oxygenase type 2-dependent prostaglandin E2 secretion is involved in retrovirus-induced T-cell dysfunction in mice
Abstract
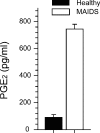
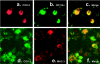
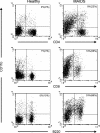
MAIDS (murine AIDS) is caused by infection with the murine leukaemia retrovirus RadLV-Rs and is characterized by a severe immunodeficiency and T-cell anergy combined with a lymphoproliferative disease affecting both B- and T-cells. Hyperactivation of the cAMP-protein kinase A pathway is involved in the T-cell dysfunction of MAIDS and HIV by inhibiting T-cell activation through the T-cell receptor. In the present study, we show that MAIDS involves a strong and selective up-regulation of cyclo-oxygenase type 2 in the CD11b+ subpopulation of T- and B-cells of the lymph nodes, leading to increased levels of PGE2 (prostaglandin E2). PGE2 activates the cAMP pathway through G-protein-coupled receptors. Treatment with cyclo-oxygenase type 2 inhibitors reduces the level of PGE2 and thereby reverses the T-cell anergy, restores the T-cell immune function and ameliorates the lymphoproliferative disease.
Figures




Similar articles
-
Mice with disrupted type I protein kinase A anchoring in T cells resist retrovirus-induced immunodeficiency.J Immunol. 2011 May 1;186(9):5119-30. doi: 10.4049/jimmunol.1100003. Epub 2011 Mar 23. J Immunol. 2011. PMID: 21430226
-
Role of cyclo-oxygenase-2 induction in interleukin-1beta induced attenuation of cultured human airway smooth muscle cell cyclic AMP generation in response to isoprenaline.Br J Pharmacol. 1998 Nov;125(6):1320-8. doi: 10.1038/sj.bjp.0702193. Br J Pharmacol. 1998. PMID: 9863663 Free PMC article.
-
Tumor cyclooxygenase-2/prostaglandin E2-dependent promotion of FOXP3 expression and CD4+ CD25+ T regulatory cell activities in lung cancer.Cancer Res. 2005 Jun 15;65(12):5211-20. doi: 10.1158/0008-5472.CAN-05-0141. Cancer Res. 2005. PMID: 15958566
-
Cells and cytokines in the pathogenesis of MAIDS, a retrovirus-induced immunodeficiency syndrome of mice.Springer Semin Immunopathol. 1995;17(2-3):231-45. doi: 10.1007/BF00196167. Springer Semin Immunopathol. 1995. PMID: 8571170 Review. No abstract available.
-
T-helper cell intrinsic defects in lupus that break peripheral tolerance to nuclear autoantigens.J Mol Med (Berl). 2005 Apr;83(4):267-78. doi: 10.1007/s00109-004-0624-2. Epub 2005 Jan 4. J Mol Med (Berl). 2005. PMID: 15630591 Review.
Cited by
-
The effect of HIV and HPV coinfection on cervical COX-2 expression and systemic prostaglandin E2 levels.Cancer Prev Res (Phila). 2012 Jan;5(1):34-40. doi: 10.1158/1940-6207.CAPR-11-0496. Epub 2011 Dec 1. Cancer Prev Res (Phila). 2012. PMID: 22135046 Free PMC article.
-
Pro- and anti-inflammatory bioactive lipids imbalance contributes to the pathobiology of autoimmune diseases.Eur J Clin Nutr. 2023 Jun;77(6):637-651. doi: 10.1038/s41430-022-01173-8. Epub 2022 Jun 14. Eur J Clin Nutr. 2023. PMID: 35701524 Review.
-
Cyclooxygenase-2 regulates Th17 cell differentiation during allergic lung inflammation.Am J Respir Crit Care Med. 2011 Jul 1;184(1):37-49. doi: 10.1164/rccm.201010-1637OC. Epub 2011 Apr 7. Am J Respir Crit Care Med. 2011. PMID: 21474648 Free PMC article.
-
EP2 receptor antagonism reduces peripheral and central hyperalgesia in a preclinical mouse model of endometriosis.Sci Rep. 2017 Mar 10;7:44169. doi: 10.1038/srep44169. Sci Rep. 2017. PMID: 28281561 Free PMC article.
-
Proinflammatory and immunoregulatory roles of eicosanoids in T cells.Front Immunol. 2013 Jun 4;4:130. doi: 10.3389/fimmu.2013.00130. eCollection 2013. Front Immunol. 2013. PMID: 23760108 Free PMC article.
References
-
- Aziz D. C., Hanna Z., Jolicoeur P. Severe immunodeficiency disease induced by a defective murine leukaemia virus. Nature (London) 1989;338:505–508. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

