Conductance studies on trichotoxin_A50E and implications for channel structure
- PMID: 15345549
- PMCID: PMC1304575
- DOI: 10.1529/biophysj.104.040659
Conductance studies on trichotoxin_A50E and implications for channel structure
Abstract
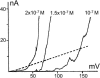
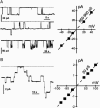
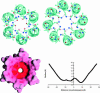
Trichotoxin_A50E is an 18-residue peptaibol whose crystal structure has recently been determined. In this study, the conductance properties of trichotoxin_A50E have been investigated in neutral planar lipid bilayers. The macroscopic current-voltage curves disclose a moderate voltage-sensitivity and the concentration-dependence suggests the channels are primarily hexameric. Under ion gradients, shifts of the reversal potential indicate that cations are preferentially transported. Trichotoxin displays only one single-channel conductance state in a given experiment, but an ensemble of experiments reveals a distribution of conductance levels. This contrasts with the related peptaibol alamethicin, which produces multiple channel levels in a single experiment, indicative of recruitment of additional monomers into different multimeric-sized channels. Based on these conductance measurements and on the recently available crystal structure of trichotoxin_A50E, which is a shorter and straighter helix than alamethicin, a tightly-packed hexameric model structure has been constructed for the trichotoxin channel. It has molecular dimensions and surface electrostatic potential compatible with the observed conductance properties of the most probable and longer-lived channel.
Figures

Similar articles
-
Model for a helical bundle channel based on the high-resolution crystal structure of trichotoxin_A50E.Biochemistry. 2002 Oct 29;41(43):12934-41. doi: 10.1021/bi026150z. Biochemistry. 2002. PMID: 12390019
-
Helical kink and channel behaviour: a comparative study with the peptaibols alamethicin, trichotoxin and antiamoebin.Eur Biophys J. 2004 May;33(3):169-74. doi: 10.1007/s00249-003-0383-y. Epub 2004 Mar 11. Eur Biophys J. 2004. PMID: 15014907 Review.
-
The properties of ion channels formed by zervamicins.Eur Biophys J. 1992;21(2):117-28. doi: 10.1007/BF00185426. Eur Biophys J. 1992. PMID: 1382967
-
Alamethicin and related peptaibols--model ion channels.Eur Biophys J. 1993;22(2):105-24. doi: 10.1007/BF00196915. Eur Biophys J. 1993. PMID: 7689461
-
Alamethicin: a peptide model for voltage gating and protein-membrane interactions.Annu Rev Biophys Biomol Struct. 1994;23:141-65. doi: 10.1146/annurev.bb.23.060194.001041. Annu Rev Biophys Biomol Struct. 1994. PMID: 7522664 Review. No abstract available.
Cited by
-
Electrophysiological Properties from Computations at a Single Voltage: Testing Theory with Stochastic Simulations.Entropy (Basel). 2021 May 6;23(5):571. doi: 10.3390/e23050571. Entropy (Basel). 2021. PMID: 34066581 Free PMC article.
-
Multi-oligomeric states of alamethicin ion channel: Assemblies and conductance.Biophys J. 2023 Jun 20;122(12):2531-2543. doi: 10.1016/j.bpj.2023.05.006. Epub 2023 May 8. Biophys J. 2023. PMID: 37161094 Free PMC article.
-
Peptaibol Production and Characterization from Trichoderma asperellum and Their Action as Biofungicide.J Fungi (Basel). 2022 Sep 29;8(10):1037. doi: 10.3390/jof8101037. J Fungi (Basel). 2022. PMID: 36294602 Free PMC article.
-
Antimicrobial peptaibols, novel suppressors of tumor cells, targeted calcium-mediated apoptosis and autophagy in human hepatocellular carcinoma cells.Mol Cancer. 2010 Feb 2;9:26. doi: 10.1186/1476-4598-9-26. Mol Cancer. 2010. PMID: 20122248 Free PMC article.
-
Validity of the Electrodiffusion Model for Calculating Conductance of Simple Ion Channels.J Phys Chem B. 2017 Apr 20;121(15):3607-3619. doi: 10.1021/acs.jpcb.6b09598. Epub 2016 Dec 12. J Phys Chem B. 2017. PMID: 27936743 Free PMC article.
References
-
- Anders, R., O. Ohlenschlager, V. Soskic, H. Wenschuh, B. Heise, and L. R. Brown. 2000. The NMR solution structure of the ion channel peptaibol chrysospermin C bound to dodecylphosphocholine micelles. Eur. J. Biochem. 267:1784–1794. - PubMed
-
- Baumann, G., and P. Mueller. 1974. A molecular model of membrane excitability. J. Supramol. Struct. 2:538–557. - PubMed
-
- Béven, L., O. Helluin, G. Molle, H. Duclohier, and H. Wróblewski. 1999. Correlation between antibacterial activity and pore-sizes of two classes of voltage-dependent channel-forming peptides. Biochim. Biophys. Acta. 1421:53–63. - PubMed
-
- Boheim, G. 1974. Statistical analysis of alamethicin channels in black lipid membranes. J. Membr. Biol. 19:277–303. - PubMed
-
- Brückner, H. 1987. Chemical synthesis of trichotoxin, a membrane active polypeptide mycotoxin. In Peptides, Proc. 19th Eur. Pept. Symp. D. Theodoropoulos, editor. De Gruyter, Berlin. 231–234.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

