The extracellular matrix gene Frem1 is essential for the normal adhesion of the embryonic epidermis
- PMID: 15345741
- PMCID: PMC518794
- DOI: 10.1073/pnas.0402760101
The extracellular matrix gene Frem1 is essential for the normal adhesion of the embryonic epidermis
Abstract
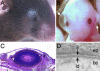
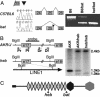
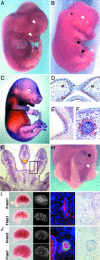
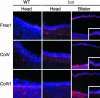
Fraser syndrome is a rare recessive disorder characterized by cryptophthalmos, syndactyly, renal defects, and a range of other developmental abnormalities. Because of their extensive phenotypic overlap, the mouse blebbing mutants have been considered models of this disorder, and the recent isolation of mutations in Fras1 in both the blebbed mouse and human Fraser patients confirms this hypothesis. Here we report the identification of mutations in an extracellular matrix gene Fras1-related extracellular matrix gene 1 (Frem1) in both the classic head blebs mutant and in an N-ethyl-N-nitrosourea-induced allele. We show that inactivation of the gene results in the formation of in utero epidermal blisters beneath the lamina densa of the basement membrane and also in renal agenesis. Frem1 is expressed widely in the developing embryo in regions of epithelial/mesenchymal interaction and epidermal remodeling. Furthermore, Frem1 appears to act as a dermal mediator of basement membrane adhesion, apparently independently of the other known "blebs" proteins Fras1 and Grip1. Unlike both Fras1 and Grip1 mutants, collagen VI and Fras1 deposition in the basement membrane is normal, indicating that the protein plays an independent role in epidermal differentiation and is required for epidermal adhesion during embryonic development.
Figures

Similar articles
-
Overlapping and divergent localization of Frem1 and Fras1 and its functional implications during mouse embryonic development.Exp Cell Res. 2007 Mar 10;313(5):910-20. doi: 10.1016/j.yexcr.2006.12.008. Epub 2006 Dec 23. Exp Cell Res. 2007. PMID: 17240369
-
The role of Fras1/Frem proteins in the structure and function of basement membrane.Int J Biochem Cell Biol. 2011 Apr;43(4):487-95. doi: 10.1016/j.biocel.2010.12.016. Epub 2010 Dec 21. Int J Biochem Cell Biol. 2011. PMID: 21182980 Review.
-
Basement membrane localization of Frem3 is independent of the Fras1/Frem1/Frem2 protein complex within the sublamina densa.Matrix Biol. 2007 Oct;26(8):652-8. doi: 10.1016/j.matbio.2007.05.008. Epub 2007 Jun 6. Matrix Biol. 2007. PMID: 17596926
-
Breakdown of the reciprocal stabilization of QBRICK/Frem1, Fras1, and Frem2 at the basement membrane provokes Fraser syndrome-like defects.Proc Natl Acad Sci U S A. 2006 Aug 8;103(32):11981-6. doi: 10.1073/pnas.0601011103. Epub 2006 Jul 31. Proc Natl Acad Sci U S A. 2006. PMID: 16880404 Free PMC article.
-
Let's stick together: the role of the Fras1 and Frem proteins in epidermal adhesion.IUBMB Life. 2007 Jul;59(7):427-35. doi: 10.1080/15216540701510581. IUBMB Life. 2007. PMID: 17654118 Review.
Cited by
-
Frem1 activity is regulated by Sonic hedgehog signaling in the cranial neural crest mesenchyme during midfacial morphogenesis.Dev Dyn. 2023 Apr;252(4):483-494. doi: 10.1002/dvdy.555. Epub 2022 Dec 19. Dev Dyn. 2023. PMID: 36495293 Free PMC article.
-
The contribution of branching morphogenesis to kidney development and disease.Nat Rev Nephrol. 2016 Dec;12(12):754-767. doi: 10.1038/nrneph.2016.157. Epub 2016 Nov 7. Nat Rev Nephrol. 2016. PMID: 27818506 Review.
-
Natural Immunity against HIV-1: Progression of Understanding after Association Studies.Viruses. 2022 Jun 8;14(6):1243. doi: 10.3390/v14061243. Viruses. 2022. PMID: 35746714 Free PMC article. Review.
-
Segmental and restricted localization pattern of Fras1 in the developing meningeal basement membrane in mouse.Histochem Cell Biol. 2013 Nov;140(5):595-601. doi: 10.1007/s00418-013-1150-5. Epub 2013 Oct 8. Histochem Cell Biol. 2013. PMID: 24101214
-
PDGFRβ regulates craniofacial development through homodimers and functional heterodimers with PDGFRα.Genes Dev. 2016 Nov 1;30(21):2443-2458. doi: 10.1101/gad.288746.116. Epub 2016 Nov 17. Genes Dev. 2016. PMID: 27856617 Free PMC article.
References
-
- Winter, R. M. (1990) Clin. Genet. 37, 494–495. - PubMed
-
- McGregor, L., Makela, V., Darling, S. M., Vrontou, S., Chalepakis, G., Roberts, C., Smart, N., Rutland, P., Prescott, N., Hopkins, J., et al. (2003) Nat. Genet. 34, 203–208. - PubMed
-
- Darling, S. & Gossler, A. (1994) Clin. Dysmorphol. 3, 91–95. - PubMed
-
- Vrontou, S., Petrou, P., Meyer, B. I., Galanopoulos, V. K., Imai, K., Yanagi, M., Chowdhury, K., Scambler, P. J. & Chalepakis, G. (2003) Nat. Genet. 34, 209–214. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

