The roles of latex and the vascular bundle in morphine biosynthesis in the opium poppy, Papaver somniferum
- PMID: 15353584
- PMCID: PMC518766
- DOI: 10.1073/pnas.0405704101
The roles of latex and the vascular bundle in morphine biosynthesis in the opium poppy, Papaver somniferum
Abstract
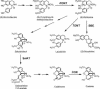
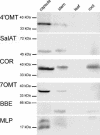
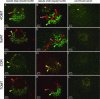
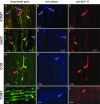
The opium poppy, Papaver somniferum, is one of mankind's oldest medicinal plants. Opium poppy today is the commercial source of the narcotic analgesics morphine and codeine. Along with these two morphinans, opium poppy produces approximately eighty alkaloids belonging to various tetrahydrobenzylisoquinoline-derived classes. It has been known for over a century that morphinan alkaloids accumulate in the latex of opium poppy. With identification of many of the enzymes of alkaloid biosynthesis in this plant, biochemical data suggested involvement of multiple cell types in alkaloid biosynthesis in poppy. Herein the immunolocalization of five enzymes of alkaloid formation in opium poppy is reported: (R,S)-3'-hydroxy-N-methylcoclaurine 4'-O-methyltransferase central to the biosynthesis of tetrahydroisoquinoline-derived alkaloids, the berberine bridge enzyme of the sanguinarine pathway, (R,S)-reticuline 7-O-methyltransferase specific to laudanosine formation, and salutaridinol 7-O-acetyltransferase and codeinone reductase, which lead to morphine. In capsule and stem, both O-methyltransferases and the O-acetyltransferase are found predominantly in parenchyma cells within the vascular bundle, and codeinone reductase is localized to laticifers, the site of morphinan alkaloid accumulation. In developing root tip, both O-methyltransferases and the O-acetyltransferase are found in the pericycle of the stele, and the berberine bridge enzyme is localized to parenchyma cells of the root cortex. Laticifers are not found in developing root tip, and, likewise, codeinone reductase was not detected. These results provide cell-specific localization that gives a coherent picture of the spatial distribution of alkaloid biosynthesis in opium poppy.
Figures
Similar articles
-
The role of phloem sieve elements and laticifers in the biosynthesis and accumulation of alkaloids in opium poppy.Plant J. 2006 Aug;47(4):547-63. doi: 10.1111/j.1365-313X.2006.02801.x. Epub 2006 Jun 30. Plant J. 2006. PMID: 16813579
-
Developmental and inducible accumulation of gene transcripts involved in alkaloid biosynthesis in opium poppy.Phytochemistry. 2003 Sep;64(1):177-86. doi: 10.1016/s0031-9422(03)00292-9. Phytochemistry. 2003. PMID: 12946416
-
A tale of three cell types: alkaloid biosynthesis is localized to sieve elements in opium poppy.Plant Cell. 2003 Nov;15(11):2626-35. doi: 10.1105/tpc.015396. Epub 2003 Sep 24. Plant Cell. 2003. PMID: 14508000 Free PMC article.
-
Benzylisoquinoline alkaloid biosynthesis in opium poppy.Planta. 2014 Jul;240(1):19-32. doi: 10.1007/s00425-014-2056-8. Epub 2014 Mar 27. Planta. 2014. PMID: 24671624 Review.
-
Evolution of morphine biosynthesis in opium poppy.Phytochemistry. 2009 Oct-Nov;70(15-16):1696-707. doi: 10.1016/j.phytochem.2009.07.006. Epub 2009 Aug 6. Phytochemistry. 2009. PMID: 19665152 Review.
Cited by
-
The role of proteomics in progressing insights into plant secondary metabolism.Front Plant Sci. 2015 Jul 7;6:504. doi: 10.3389/fpls.2015.00504. eCollection 2015. Front Plant Sci. 2015. PMID: 26217358 Free PMC article. Review.
-
Comparative macroarray analysis of morphine containing Papaver somniferum and eight morphine free Papaver species identifies an O-methyltransferase involved in benzylisoquinoline biosynthesis.Planta. 2005 Oct;222(3):458-71. doi: 10.1007/s00425-005-1550-4. Epub 2005 Jul 21. Planta. 2005. PMID: 16034588
-
Tempo-Spatial Pattern of Stepharine Accumulation in Stephania Glabra Morphogenic Tissues.Int J Mol Sci. 2019 Feb 13;20(4):808. doi: 10.3390/ijms20040808. Int J Mol Sci. 2019. PMID: 30781887 Free PMC article.
-
Identification of a Second Site of Pyrrolizidine Alkaloid Biosynthesis in Comfrey to Boost Plant Defense in Floral Stage.Plant Physiol. 2017 May;174(1):47-55. doi: 10.1104/pp.17.00265. Epub 2017 Mar 8. Plant Physiol. 2017. PMID: 28275146 Free PMC article.
-
The genetic manipulation of medicinal and aromatic plants.Plant Cell Rep. 2007 Oct;26(10):1689-715. doi: 10.1007/s00299-007-0384-x. Epub 2007 Jul 3. Plant Cell Rep. 2007. PMID: 17609957 Review.
References
-
- Kutchan, T. M., Ayabe, S. & Coscia, C. J. (1985) in The Chemistry and Biology of Isoquinoline Alkaloids, eds. Phillipson, J. D., Roberts, M. F. & Zenk, M. H. (Springer, Berlin), pp. 281–294.
-
- Felkova, M. & Babkova, K. (1958) Pharmazie 13, 220. - PubMed
-
- Fairbairn, J. W. & Kapoor, L. D. (1960) Planta Med. 8, 49–61.
-
- Nessler, C. L. & Mahlberg, P. G. (1978) Am. J. Bot. 65, 978–983.
-
- Roberts, M. F., McCarthy, D., Kutchan, T. M. & Coscia, C. J. (1983) Arch. Biochem. Biophys. 222, 599–609. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

