Synergistic interactions of lipids and myelin basic protein
- PMID: 15353595
- PMCID: PMC518780
- DOI: 10.1073/pnas.0405665101
Synergistic interactions of lipids and myelin basic protein
Abstract
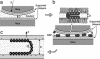
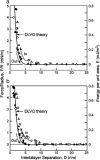
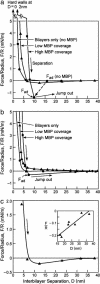
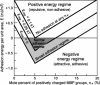
This report describes force measurements and atomic force microscope imaging of lipid-protein interactions that determine the structure of a model membrane system that closely mimics the myelin sheath. Our results suggest that noncovalent, mainly electrostatic and hydrophobic, interactions are responsible for the multilamellar structure and stability of myelin. We find that myelin basic protein acts as a lipid coupler between two apposed bilayers and as a lipid "hole-filler," effectively preventing defect holes from developing. From our protein-mediated-adhesion and force-distance measurements, we develop a simple quantitative model that gives a reasonably accurate picture of the molecular mechanism and adhesion of bilayer-bridging proteins by means of noncovalent interactions. The results and model indicate that optimum myelin adhesion and stability depend on the difference between, rather than the product of, the opposite charges on the lipid bilayers and myelin basic protein, as well as on the repulsive forces associated with membrane fluidity, and that small changes in any of these parameters away from the synergistically optimum values can lead to large changes in the adhesion or even its total elimination. Our results also show that the often-asked question of which membrane species, the lipids or the proteins, are the "important ones" may be misplaced. Both components work synergistically to provide the adhesion and overall structure. A better appreciation of the mechanism of this synergy may allow for a better understanding of stacked and especially myelin membrane structures and may lead to better treatments for demyelinating diseases such as multiple sclerosis.
Figures

Similar articles
-
Interaction forces and adhesion of supported myelin lipid bilayers modulated by myelin basic protein.Proc Natl Acad Sci U S A. 2009 Mar 3;106(9):3154-9. doi: 10.1073/pnas.0813110106. Epub 2009 Feb 13. Proc Natl Acad Sci U S A. 2009. PMID: 19218452 Free PMC article.
-
Lipid domains control myelin basic protein adsorption and membrane interactions between model myelin lipid bilayers.Proc Natl Acad Sci U S A. 2014 Feb 25;111(8):E768-75. doi: 10.1073/pnas.1401165111. Epub 2014 Feb 10. Proc Natl Acad Sci U S A. 2014. PMID: 24516125 Free PMC article.
-
Force measurements on myelin basic protein adsorbed to mica and lipid bilayer surfaces done with the atomic force microscope.Biophys J. 1999 Feb;76(2):1072-9. doi: 10.1016/S0006-3495(99)77272-3. Biophys J. 1999. PMID: 9916039 Free PMC article.
-
Central nervous system myelin: structure, function, and pathology.Clin Biochem. 1991 Apr;24(2):113-34. doi: 10.1016/0009-9120(91)90421-a. Clin Biochem. 1991. PMID: 1710177 Free PMC article. Review.
-
Oligodendroglial membrane dynamics in relation to myelin biogenesis.Cell Mol Life Sci. 2016 Sep;73(17):3291-310. doi: 10.1007/s00018-016-2228-8. Epub 2016 May 3. Cell Mol Life Sci. 2016. PMID: 27141942 Free PMC article. Review.
Cited by
-
Interfacial rheology and direct imaging reveal domain-templated network formation in phospholipid monolayers penetrated by fibrinogen.Soft Matter. 2019 Nov 28;15(44):9076-9084. doi: 10.1039/c9sm01519a. Epub 2019 Oct 25. Soft Matter. 2019. PMID: 31651923 Free PMC article.
-
Inhibition of pulmonary surfactant adsorption by serum and the mechanisms of reversal by hydrophilic polymers: theory.Biophys J. 2005 Sep;89(3):1621-9. doi: 10.1529/biophysj.105.062646. Epub 2005 Jul 8. Biophys J. 2005. PMID: 16006630 Free PMC article.
-
Flexible Players within the Sheaths: The Intrinsically Disordered Proteins of Myelin in Health and Disease.Cells. 2020 Feb 18;9(2):470. doi: 10.3390/cells9020470. Cells. 2020. PMID: 32085570 Free PMC article. Review.
-
Critical and off-critical miscibility transitions in model extracellular and cytoplasmic myelin lipid monolayers.Biophys J. 2011 Mar 16;100(6):1490-8. doi: 10.1016/j.bpj.2011.02.009. Biophys J. 2011. PMID: 21402031 Free PMC article.
-
Mussel-designed protective coatings for compliant substrates.J Dent Res. 2008 Aug;87(8):701-9. doi: 10.1177/154405910808700808. J Dent Res. 2008. PMID: 18650539 Free PMC article. Review.
References
-
- Morell, P. & Norton, W. T. (1980) Sci. Am. 242, 88–90. - PubMed
-
- Boggs, J. M. & Moscarello, M. A. (1978) Biochim. Biophys. Acta 515, 1–21. - PubMed
-
- Wood, D. D. & Moscarello, M. A. (1984) J. Membr. Biol. 79, 195–201. - PubMed
-
- Cuzner, M. L. & Norton, W. T. (1996) Brain Pathol. 6, 231–242. - PubMed
-
- Genain, C. P., Cannella, B., Hauser, S. L. & Raine, C. S. (1999) Nat. Med. 5, 170–175. - PubMed

