Generation of reactive oxygen species in the reaction catalyzed by alpha-ketoglutarate dehydrogenase
- PMID: 15356188
- PMCID: PMC6729921
- DOI: 10.1523/JNEUROSCI.1842-04.2004
Generation of reactive oxygen species in the reaction catalyzed by alpha-ketoglutarate dehydrogenase
Abstract
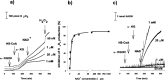
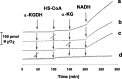
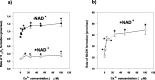
Alpha-ketoglutarate dehydrogenase (alpha-KGDH), a key enzyme in the Krebs' cycle, is a crucial early target of oxidative stress (Tretter and Adam-Vizi, 2000). The present study demonstrates that alpha-KGDH is able to generate H(2)O(2) and, thus, could also be a source of reactive oxygen species (ROS) in mitochondria. Isolated alpha-KGDH with coenzyme A (HS-CoA) and thiamine pyrophosphate started to produce H(2)O(2) after addition of alpha-ketoglutarate in the absence of nicotinamide adenine dinucleotide-oxidized (NAD(+)). NAD(+), which proved to be a powerful inhibitor of alpha-KGDH-mediated H(2)O(2) formation, switched the H(2)O(2) forming mode of the enzyme to the catalytic [nicotinamide adenine dinucleotide-reduced (NADH) forming] mode. In contrast, NADH stimulated H(2)O(2) formation by alpha-KGDH, and for this, neither alpha-ketoglutarate nor HS-CoA were required. When all of the substrates and cofactors of the enzyme were present, the NADH/NAD(+) ratio determined the rate of H(2)O(2) production. The higher the NADH/NAD(+) ratio the higher the rate of H(2)O(2) production. H(2)O(2) production as well as the catalytic function of the enzyme was activated by Ca(2+). In synaptosomes, using alpha-ketoglutarate as respiratory substrate, the rate of H(2)O(2) production increased by 2.5-fold, and aconitase activity decreased, indicating that alpha-KGDH can generate H(2)O(2) in in situ mitochondria. Given the NADH/NAD(+) ratio as a key regulator of H(2)O(2) production by alpha-KGDH, it is suggested that production of ROS could be significant not only in the respiratory chain but also in the Krebs' cycle when oxidation of NADH is impaired. Thus alpha-KGDH is not only a target of ROS but could significantly contribute to generation of oxidative stress in the mitochondria.
Figures




References
-
- Andersson U, Leighton B, Young ME, Blomstrand E, Newsholme EA (1998) Inactivation of aconitase and oxoglutarate dehydrogenase in skeletal muscle in vitro by superoxide anions and/or nitric oxide. Biochem Biophys Res Commun 249: 512-516. - PubMed
-
- Badwey JA, Karnovsky ML (1979) Production of superoxide and hydrogen peroxide by an NADH-oxidase in guinea pig polymorphonuclear leukocytes. Modulation by nucleotides and divalent cations. J Biol Chem 254: 11530-11537. - PubMed
-
- Beal MF (1996) Mitochondria, free radicals, and neurodegeneration. Curr Opin Neurobiol 6: 661-666. - PubMed
-
- Betarbet R, Sherer TB, MacKenzie G, Garcia-Osuna M, Panov AV, Greenamyre JT (2000) Chronic systemic pesticide exposure reproduces features of Parkinson's disease. Nat Neurosci 3: 1301-1306. - PubMed
-
- Bindoli A (1988) Lipid peroxidation in mitochondria. Free Radic Biol Med 5: 247-261. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
