Interactions between the innate immune and blood coagulation systems
- PMID: 15364056
- PMCID: PMC7185622
- DOI: 10.1016/j.it.2004.08.003
Interactions between the innate immune and blood coagulation systems
Abstract
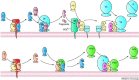
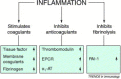
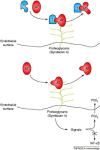
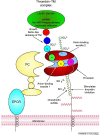
Blood coagulation and inflammation are universal responses to infection and there is crosstalk between inflammation and coagulation that can either amplify or dampen the responses. Loss of appropriate interactions between these systems probably contributes to morbidity and mortality in infectious diseases. For instance, inflammatory cytokines and leukocyte elastase can downregulate natural anticoagulant proteins that help to maintain endothelial-cell integrity, control clotting, inhibit vasoactive peptides and dampen leukocyte infiltration into the vessel wall. This Review will summarize our current understanding of the mechanisms involved in the crosstalk between these two important systems.
Figures

Similar articles
-
The urokinase receptor system, a key regulator at the intersection between inflammation, immunity, and coagulation.Curr Pharm Des. 2011;17(19):1924-43. doi: 10.2174/138161211796718189. Curr Pharm Des. 2011. PMID: 21711238 Review.
-
The reciprocal relationship between inflammation and coagulation.Top Companion Anim Med. 2012 May;27(2):46-52. doi: 10.1053/j.tcam.2012.06.003. Epub 2012 Aug 2. Top Companion Anim Med. 2012. PMID: 23031455 Review.
-
Interactions between coagulation and inflammation.Scand J Infect Dis. 2003;35(9):545-54. doi: 10.1080/00365540310015638. Scand J Infect Dis. 2003. PMID: 14620133 Review.
-
A novel C5a receptor-tissue factor cross-talk in neutrophils links innate immunity to coagulation pathways.J Immunol. 2006 Oct 1;177(7):4794-802. doi: 10.4049/jimmunol.177.7.4794. J Immunol. 2006. PMID: 16982920
-
Dangerous liaisons: complement, coagulation, and kallikrein/kinin cross-talk act as a linchpin in the events leading to thromboinflammation.Immunol Rev. 2016 Nov;274(1):245-269. doi: 10.1111/imr.12471. Immunol Rev. 2016. PMID: 27782319 Review.
Cited by
-
Unexpected role of anticoagulant protein C in controlling epithelial barrier integrity and intestinal inflammation.Proc Natl Acad Sci U S A. 2011 Dec 6;108(49):19830-5. doi: 10.1073/pnas.1107140108. Epub 2011 Nov 22. Proc Natl Acad Sci U S A. 2011. PMID: 22109555 Free PMC article.
-
Whole genome bisulfite sequencing reveals DNA methylation roles in the adaptive response of wildness training giant pandas to wild environment.Front Genet. 2022 Oct 11;13:995700. doi: 10.3389/fgene.2022.995700. eCollection 2022. Front Genet. 2022. PMID: 36303550 Free PMC article.
-
Matrix metalloproteinase-9, -10, and tissue inhibitor of matrix metalloproteinases-1 blood levels as biomarkers of severity and mortality in sepsis.Crit Care. 2009;13(5):R158. doi: 10.1186/cc8115. Epub 2009 Oct 2. Crit Care. 2009. PMID: 19799791 Free PMC article.
-
Computational identification and clinical validation of a novel risk signature based on coagulation-related lncRNAs for predicting prognosis, immunotherapy response, and chemosensitivity in colorectal cancer patients.Front Immunol. 2023 Oct 19;14:1279789. doi: 10.3389/fimmu.2023.1279789. eCollection 2023. Front Immunol. 2023. PMID: 37928532 Free PMC article.
-
Molecular intercommunication between the complement and coagulation systems.J Immunol. 2010 Nov 1;185(9):5628-36. doi: 10.4049/jimmunol.0903678. Epub 2010 Sep 24. J Immunol. 2010. PMID: 20870944 Free PMC article.
References
-
- Opal S.M. Systemic host responses in severe sepsis analyzed by causative microorganism and treatment effects of drotrecogin alfa (activated) Clin. Infect. Dis. 2003;37:50–58. - PubMed
-
- Lee N. A major outbreak of severe acute respiratory syndrome in Hong Kong. N. Engl. J. Med. 2003;348:1986–1994. - PubMed
-
- Rauch U. Transfer of tissue factor from leukocytes to platelets is mediated by CD15 and tissue factor. Blood. 2000;96:170–175. - PubMed
-
- Bevers E.M. Transmembrane phospholipid distribution in blood cells: control mechanisms and pathophysiological significance. Biol. Chem. 1998;379:973–986. - PubMed
-
- Sims P.J. Assembly of the platelet prothrombinase complex is linked to vesiculation of the platelet plasma membrane. Studies in Scott Syndrome: An isolated defect in platelet procoagulant activity. J. Biol. Chem. 1989;264:17049–17057. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

