Human cytomegalovirus inhibits cytokine-induced macrophage differentiation
- PMID: 15367604
- PMCID: PMC516431
- DOI: 10.1128/JVI.78.19.10378-10389.2004
Human cytomegalovirus inhibits cytokine-induced macrophage differentiation
Abstract
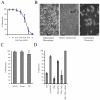
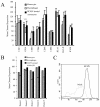
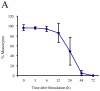
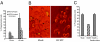
Human cytomegalovirus (HCMV) infection in immunocompromised patients is associated with impaired immunological function. Blood monocytes, which differentiate into macrophage effector cells, are of central importance for immune reactivity. Here, we demonstrate that HCMV transiently blocks cytokine-induced differentiation of monocytes into functionally active phagocytic macrophages. In HCMV-treated cultures, the cells had classical macrophage markers but lacked the classical morphological appearance of macrophages and had impairments in migration and phagocytosis. Even at very low multiplicities of infection, macrophage differentiation was almost completely inhibited. The inhibition appeared to be mediated by a soluble factor released upon viral treatment of monocytes. Human immunodeficiency virus or measles virus had no such effects. These findings suggest that HCMV impairs immune function by blocking certain aspects of cytokine-induced differentiation of monocytes and demonstrate an efficient pathway for this virus to evade immune recognition that may have clinical implications for the generalized immunosuppression often observed in HCMV-infected patients.
Figures


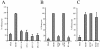
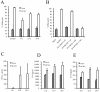
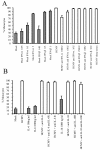
Similar articles
-
Growth of human cytomegalovirus in primary macrophages.Methods. 1998 Sep;16(1):126-38. doi: 10.1006/meth.1998.0650. Methods. 1998. PMID: 9774522
-
Human cytomegalovirus inhibits differentiation of monocytes into dendritic cells with the consequence of depressed immunological functions.J Virol. 2003 Oct;77(20):10943-56. doi: 10.1128/jvi.77.20.10943-10956.2003. J Virol. 2003. PMID: 14512544 Free PMC article.
-
Human macrophage differentiation in vivo and in vitro. A comparison of human peritoneal macrophages and monocytes.Acta Pathol Microbiol Scand C. 1979 Apr;87C(2):113-20. Acta Pathol Microbiol Scand C. 1979. PMID: 375676
-
Cytomegalovirus persistence in macrophages and endothelial cells.Scand J Infect Dis Suppl. 1995;99:34-40. Scand J Infect Dis Suppl. 1995. PMID: 8668941 Review.
-
Regulation of chemokine/cytokine network during in vitro differentiation and HIV-1 infection of human monocytes: possible importance in the pathogenesis of AIDS.J Leukoc Biol. 2000 Sep;68(3):391-9. J Leukoc Biol. 2000. PMID: 10985256 Review.
Cited by
-
The HCMV tegument protein UL88 degrades MyD88 and reduces innate immune activation.J Virol. 2025 Aug 19;99(8):e0041425. doi: 10.1128/jvi.00414-25. Epub 2025 Jul 10. J Virol. 2025. PMID: 40638072 Free PMC article.
-
The Differentiation of Human Cytomegalovirus Infected-Monocytes Is Required for Viral Replication.Front Cell Infect Microbiol. 2020 Jul 31;10:368. doi: 10.3389/fcimb.2020.00368. eCollection 2020. Front Cell Infect Microbiol. 2020. PMID: 32850474 Free PMC article. Review.
-
Inflammatory Breast Cancer: The Secretome of HCMV+ Tumor-Associated Macrophages Enhances Proliferation, Invasion, Colony Formation, and Expression of Cancer Stem Cell Markers.Front Oncol. 2022 Jun 30;12:899622. doi: 10.3389/fonc.2022.899622. eCollection 2022. Front Oncol. 2022. PMID: 35847899 Free PMC article.
-
Is Pulmonary non-Tuberculous Mycobacterial Disease Linked with a High Burden of Latent Cytomegalovirus?J Clin Immunol. 2016 Feb;36(2):113-6. doi: 10.1007/s10875-016-0233-1. Epub 2016 Jan 13. J Clin Immunol. 2016. PMID: 26759253
-
Dendritic cell biology in human cytomegalovirus infection and the clinical consequences for host immunity and pathology.Virulence. 2012 Nov 15;3(7):621-34. doi: 10.4161/viru.22239. Epub 2012 Oct 17. Virulence. 2012. PMID: 23076329 Free PMC article. Review.
References
-
- Allavena, P., L. Piemonti, D. Longoni, S. Bernasconi, A. Stoppacciaro, L. Ruco, and A. Mantovani. 1998. IL-10 prevents the differentiation of monocytes to dendritic cells but promotes their maturation to macrophages. Eur. J. Immunol. 28:359-369. - PubMed
-
- Asadullah, K., S. Prosch, H. Audring, I. Buttnerova, H. D. Volk, W. Sterry, and W. D. Docke. 1999. A high prevalence of cytomegalovirus antigenaemia in patients with moderate to severe chronic plaque psoriasis: an association with systemic tumour necrosis factor alpha overexpression. Br. J. Dermatol. 141:94-102. - PubMed
-
- Biondi, A., T. H. Rossing, J. Bennett, and R. F. Todd III. 1984. Surface membrane heterogeneity among human mononuclear phagocytes. J. Immunol. 132:1237-1243. - PubMed
-
- Bolovan-Fritts, C. A., E. S. Mocarski, and J. A. Wiedeman. 1999. Peripheral blood CD14(+) cells from healthy subjects carry a circular conformation of latent cytomegalovirus genome. Blood 93:394-398. - PubMed
-
- Britt, W. J., and C. A. Alford. 1996. Cytomegalovirus, p. 2493-2523. In B. N. Fields, D. M. Knipe, and P. M. Howley (ed.), Fields virology, vol. 2, 3rd ed. Lippincott-Raven Publishers, Philadelphia, Pa.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

