Early embryonic death in mice lacking the beta-catenin-binding protein Duplin
- PMID: 15367660
- PMCID: PMC516734
- DOI: 10.1128/MCB.24.19.8386-8394.2004
Early embryonic death in mice lacking the beta-catenin-binding protein Duplin
Abstract
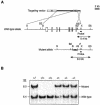
The Wnt signaling pathway plays a pivotal role in vertebrate early development and morphogenesis. Duplin (axis duplication inhibitor) interacts with beta-catenin and prevents its binding to Tcf, thereby inhibiting downstream Wnt signaling. Here we show that Duplin is expressed predominantly from early- to mid-stage mouse embryogenesis, and we describe the generation of mice deficient in Duplin. Duplin(-/-) embryos manifest growth retardation from embryonic day 5.5 (E5.5) and developmental arrest accompanied by massive apoptosis at E7.5. The mutant embryos develop into an egg cylinder but do not form a primitive streak or mesoderm. Expression of beta-catenin target genes, including those for T (brachyury), Axin2, and cyclin D1, was not increased in Duplin(-/-) embryos, suggesting that the developmental defect is not simply attributable to upregulation of Wnt signaling caused by the lack of this inhibitor. These results suggest that Duplin plays an indispensable role, likely by a mechanism independent of inhibition of Wnt signaling, in mouse embryonic growth and differentiation at an early developmental stage.
Figures




Similar articles
-
Beta-catenin signaling marks the prospective site of primitive streak formation in the mouse embryo.Dev Dyn. 2004 Oct;231(2):416-24. doi: 10.1002/dvdy.20135. Dev Dyn. 2004. PMID: 15366019
-
Nuclear localization of Duplin, a beta-catenin-binding protein, is essential for its inhibitory activity on the Wnt signaling pathway.J Biol Chem. 2002 Feb 22;277(8):5816-22. doi: 10.1074/jbc.M108433200. Epub 2001 Dec 13. J Biol Chem. 2002. PMID: 11744694
-
Canonical Wnt signaling is required for development of embryonic stem cell-derived mesoderm.Development. 2006 Oct;133(19):3787-96. doi: 10.1242/dev.02551. Epub 2006 Aug 30. Development. 2006. PMID: 16943279
-
Quercetin inhibit human SW480 colon cancer growth in association with inhibition of cyclin D1 and survivin expression through Wnt/beta-catenin signaling pathway.Cancer Invest. 2009 Jul;27(6):604-12. doi: 10.1080/07357900802337191. Cancer Invest. 2009. PMID: 19440933
-
Suppression of STAT3 activity by Duplin, which is a negative regulator of the Wnt signal.J Biochem. 2006 Feb;139(2):305-14. doi: 10.1093/jb/mvj033. J Biochem. 2006. PMID: 16452319
Cited by
-
Oxytocin ameliorates impaired social behavior in a Chd8 haploinsufficiency mouse model of autism.BMC Neurosci. 2021 May 1;22(1):32. doi: 10.1186/s12868-021-00631-6. BMC Neurosci. 2021. PMID: 33933000 Free PMC article.
-
Sentinels of chromatin: chromodomain helicase DNA-binding proteins in development and disease.Genes Dev. 2021 Nov 1;35(21-22):1403-1430. doi: 10.1101/gad.348897.121. Genes Dev. 2021. PMID: 34725129 Free PMC article. Review.
-
Fbxw7 regulates lipid metabolism and cell fate decisions in the mouse liver.J Clin Invest. 2011 Jan;121(1):342-54. doi: 10.1172/JCI40725. Epub 2010 Dec 1. J Clin Invest. 2011. PMID: 21123947 Free PMC article.
-
Chd8 haploinsufficiency impairs early brain development and protein homeostasis later in life.Mol Autism. 2020 Oct 5;11(1):74. doi: 10.1186/s13229-020-00369-8. Mol Autism. 2020. PMID: 33023670 Free PMC article.
-
The chromatin Remodeler CHD8 is required for activation of progesterone receptor-dependent enhancers.PLoS Genet. 2015 Apr 20;11(4):e1005174. doi: 10.1371/journal.pgen.1005174. eCollection 2015 Apr. PLoS Genet. 2015. PMID: 25894978 Free PMC article.
References
-
- Arnold, S. J., J. Stappert, A. Bauer, A. Kispert, B. G. Herrmann, and R. Kemler. 2000. Brachyury is a target gene of the Wnt/β-catenin signaling pathway. Mech. Dev. 91:249-258. - PubMed
-
- Behrens, J., B. A. Jerchow, M. Wurtele, J. Grimm, C. Asbrand, R. Wirtz, M. Kuhl, D. Wedlich, and W. Birchmeier. 1998. Functional interaction of an axin homolog, conductin, with β-catenin, APC, and GSK3β. Science 280:596-599. - PubMed
-
- Bienz, M., and H. Clevers. 2000. Linking colorectal cancer to Wnt signaling. Cell 103:311-320. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
