The adaptor protein AP-4 as a component of the clathrin coat machinery: a morphological study
- PMID: 15377281
- PMCID: PMC1134722
- DOI: 10.1042/BJ20041010
The adaptor protein AP-4 as a component of the clathrin coat machinery: a morphological study
Abstract
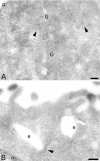
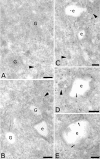
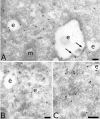
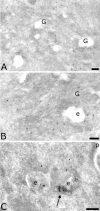
The four members of the AP (adaptor protein) family are heterotetrameric cytosolic complexes that are involved in the intracellular trafficking of cargo proteins between different organelles. They interact with motifs present in the cytoplasmic tails of their specific cargo proteins at different intracellular locations. While AP-1, AP-2 and AP-3 have been investigated extensively, very few studies have focused on the fourth member, AP-4. In the present study, we report on the intracellular localization of AP-4 in the MDCK (Madin-Darby canine kidney) and MelJuSo cell lines after immunogold labelling of ultrathin cryosections. We find that AP-4 is localized mainly in the Golgi complex, as well as on endosomes and transport vesicles. Interestingly, we show for the first time that AP-4 is localized with the clathrin coat machinery in the Golgi complex and in the endocytic pathway. Furthermore, we find that AP-4 is localized with the CI-MPR (cation-independent mannose 6-phosphate receptor), but not with the transferrin receptor, LAMP-2 (lysosomal-associated membrane protein-2) or invariant chain. The difference in morphology between CI-MPR/AP-4-positive vesicles and CI-MPR/AP-1-positive vesicles raises the possibility that AP-4 acts at a location different from that of AP-1 in the intracellular trafficking pathway of CI-MPR.
Figures



Similar articles
-
Intracellular transport of MHC class II and associated invariant chain in antigen presenting cells from AP-3-deficient mocha mice.Cell Immunol. 2001 Jun 15;210(2):143-53. doi: 10.1006/cimm.2001.1817. Cell Immunol. 2001. PMID: 11520080
-
The adaptor complex AP-2 regulates post-endocytic trafficking through the non-clathrin Arf6-dependent endocytic pathway.J Cell Sci. 2008 Dec 15;121(Pt 24):4008-17. doi: 10.1242/jcs.033522. Epub 2008 Nov 25. J Cell Sci. 2008. PMID: 19033387
-
AP-1 and AP-3 mediate sorting of melanosomal and lysosomal membrane proteins into distinct post-Golgi trafficking pathways.Traffic. 2008 Jul;9(7):1157-72. doi: 10.1111/j.1600-0854.2008.00745.x. Epub 2008 Apr 9. Traffic. 2008. PMID: 18410487
-
Neuronal and non-neuronal functions of the AP-3 sorting machinery.J Cell Sci. 2007 Feb 15;120(Pt 4):531-41. doi: 10.1242/jcs.03365. J Cell Sci. 2007. PMID: 17287392 Review.
-
Life of a clathrin coat: insights from clathrin and AP structures.Nat Rev Mol Cell Biol. 2006 Jan;7(1):32-44. doi: 10.1038/nrm1786. Nat Rev Mol Cell Biol. 2006. PMID: 16493411 Review.
Cited by
-
Characterization of the malaria parasite Plasmodium falciparum Tepsin homolog.Microbiol Spectr. 2025 Aug 5;13(8):e0328824. doi: 10.1128/spectrum.03288-24. Epub 2025 Jun 30. Microbiol Spectr. 2025. PMID: 40586818 Free PMC article.
-
UNC93B1 mediates differential trafficking of endosomal TLRs.Elife. 2013 Feb 19;2:e00291. doi: 10.7554/eLife.00291. Elife. 2013. PMID: 23426999 Free PMC article.
-
Demonstration of correlative atomic force and transmission electron microscopy using actin cytoskeleton.Biophys Physicobiol. 2017 Jul 28;14:111-117. doi: 10.2142/biophysico.14.0_111. eCollection 2017. Biophys Physicobiol. 2017. PMID: 28828286 Free PMC article.
-
Adapting for endocytosis: roles for endocytic sorting adaptors in directing neural development.Front Cell Neurosci. 2015 Apr 8;9:119. doi: 10.3389/fncel.2015.00119. eCollection 2015. Front Cell Neurosci. 2015. PMID: 25904845 Free PMC article. Review.
-
Endosomal traffic disorders: a driving force behind neurodegenerative diseases.Transl Neurodegener. 2024 Dec 24;13(1):66. doi: 10.1186/s40035-024-00460-7. Transl Neurodegener. 2024. PMID: 39716330 Free PMC article. Review.
References
-
- Boehm M., Bonifacino J. S. Genetic analyses of adaptin function from yeast to mammals. Gene. 2002;286:175–186. - PubMed
-
- Kirchhausen T. Three ways to make a vesicle. Nat. Rev. Mol. Cell Biol. 2000;1:187–198. - PubMed
-
- Bonifacino J. S., Traub L. M. Signals for sorting of transmembrane proteins to endosomes and lysosomes. Annu. Rev. Biochem. 2003;72:395–447. - PubMed
-
- Ohno H., Aguilar R. C., Yeh D., Taura D., Saito T., Bonifacino J. S. The medium subunits of adaptor complexes recognize distinct but overlapping sets of tyrosine-based sorting signals. J. Biol. Chem. 1998;273:25915–25921. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

