Checking the pH-induced conformational transition of prion protein by molecular dynamics simulations: effect of protonation of histidine residues
- PMID: 15377536
- PMCID: PMC1304876
- DOI: 10.1529/biophysj.104.043448
Checking the pH-induced conformational transition of prion protein by molecular dynamics simulations: effect of protonation of histidine residues
Abstract
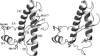
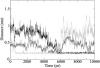
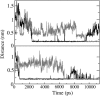
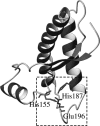
The role of acidic pH in the conversion of human prion protein to the pathogenic isoform is investigated by means of molecular dynamics simulations, focusing the attention on the effect of protonation of histidine residues on the conformational behavior of human PrPC globular domain. Our simulations reveal a significant loss of alpha-helix content under mildly acidic conditions, due to destructuration of the C-terminal part of HB (thus suggesting a possible involvement of HB into the conformational transition leading to the pathogenic isoform) and a transient lengthening of the native beta-sheet. Protonation of His-187 and His-155 seems to be crucial for the onset of the conformational rearrangement. This finding can be related to the existence of a pathogenic mutation, H187R, which is associated with GSS syndrome. Finally, the relevance of our results for the location of a Cu2+-binding pocket in the C-terminal part of the prion is discussed.
Figures



Similar articles
-
Assessing the acid-base and conformational properties of histidine residues in human prion protein (125-228) by means of pK(a) calculations and molecular dynamics simulations.Proteins. 2006 Jul 1;64(1):167-77. doi: 10.1002/prot.20979. Proteins. 2006. PMID: 16639746
-
Unveiling the role of histidine and tyrosine residues on the conformation of the avian prion hexarepeat domain.J Phys Chem B. 2008 Apr 24;112(16):5182-8. doi: 10.1021/jp710702q. Epub 2008 Apr 3. J Phys Chem B. 2008. PMID: 18386869
-
Conformational polymorphism of the amyloidogenic peptide homologous to residues 113-127 of the prion protein.Biophys J. 2003 Jul;85(1):473-83. doi: 10.1016/S0006-3495(03)74492-0. Biophys J. 2003. PMID: 12829502 Free PMC article.
-
Molecular dynamics as an approach to study prion protein misfolding and the effect of pathogenic mutations.Top Curr Chem. 2011;305:169-97. doi: 10.1007/128_2011_158. Top Curr Chem. 2011. PMID: 21526434 Review.
-
Glycosylation and prion protein.Curr Opin Struct Biol. 2002 Oct;12(5):578-86. doi: 10.1016/s0959-440x(02)00377-9. Curr Opin Struct Biol. 2002. PMID: 12464308 Review.
Cited by
-
Hydration effects on the HET-s prion and amyloid-beta fibrillous aggregates, studied with three-dimensional molecular theory of solvation.Biophys J. 2008 Nov 15;95(10):4540-8. doi: 10.1529/biophysj.107.123000. Epub 2008 Aug 8. Biophys J. 2008. PMID: 18689456 Free PMC article.
-
Partially Unfolded Forms of the Prion Protein Populated under Misfolding-promoting Conditions: CHARACTERIZATION BY HYDROGEN EXCHANGE MASS SPECTROMETRY AND NMR.J Biol Chem. 2015 Oct 16;290(42):25227-40. doi: 10.1074/jbc.M115.677575. Epub 2015 Aug 25. J Biol Chem. 2015. PMID: 26306043 Free PMC article.
-
Structural and hydration properties of the partially unfolded states of the prion protein.Biophys J. 2007 Aug 15;93(4):1284-92. doi: 10.1529/biophysj.107.108613. Epub 2007 May 4. Biophys J. 2007. PMID: 17483173 Free PMC article.
-
Searching for factors that distinguish disease-prone and disease-resistant prions via sequence analysis.Bioinform Biol Insights. 2008 Mar 12;2:133-44. doi: 10.4137/bbi.s550. Bioinform Biol Insights. 2008. PMID: 19812771 Free PMC article.
-
Probing the role of PrP repeats in conformational conversion and amyloid assembly of chimeric yeast prions.J Biol Chem. 2007 Nov 23;282(47):34204-12. doi: 10.1074/jbc.M704952200. Epub 2007 Sep 24. J Biol Chem. 2007. PMID: 17893150 Free PMC article.
References
-
- Alonso, D. O. V., C. An, and V. Daggett. 2002. Simulations of biomolecules: characterization of the early steps in the pH-induced conformational conversion of the hamster, bovine and human forms of the prion protein. Philos. Trans. R. Soc. Lond. A. 360:1165–1178. - PubMed
-
- Amadei, A., G. Chillemi, M. A. Ceruso, A. Grottesi, and A. Di Nola. 2000. Molecular dynamics simulations with constrained roto-translational motions: Theoretical basis and statistical mechanical consistency. J. Chem. Phys. 112:9–23.
-
- Apetri, A. C., and W. K. Surewicz. 2002. Kinetic intermediate in the folding of human prion protein. J. Biol. Chem. 277:44589–44592. - PubMed
-
- Bashford, D. and K. Gerwert. 1992. Electrostatic calculations of the pKa values of ionizable groups in bacteriorhodopsin. J. Mol. Biol. 224:473–486. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

