Protection against Plasmodium chabaudi malaria induced by immunization with apical membrane antigen 1 and merozoite surface protein 1 in the absence of gamma interferon or interleukin-4
- PMID: 15385457
- PMCID: PMC517570
- DOI: 10.1128/IAI.72.10.5605-5612.2004
Protection against Plasmodium chabaudi malaria induced by immunization with apical membrane antigen 1 and merozoite surface protein 1 in the absence of gamma interferon or interleukin-4
Abstract
Strategies to optimize formulations of multisubunit malaria vaccines require a basic knowledge of underlying protective immune mechanisms induced by each vaccine component. In the present study, we evaluated the contribution of antibody-mediated and cell-mediated immune mechanisms to the protection induced by immunization with two blood-stage malaria vaccine candidate antigens, apical membrane antigen 1 (AMA-1) and merozoite surface protein 1 (MSP-1). Immunologically intact or selected immunologic knockout mice were immunized with purified recombinant Plasmodium chabaudi AMA-1 (PcAMA-1) and/or the 42-kDa C-terminal processing fragment of P. chabaudi MSP-1 (MSP-1(42)). The efficacy of immunization in each animal model was measured as protection against blood-stage P. chabaudi malaria. Immunization of B-cell-deficient JH(-/-) mice indicated that PcAMA-1 vaccine-induced immunity is largely antibody dependent. In contrast, JH(-/-) mice immunized with PcMSP-1(42) were partially protected against P. chabaudi malaria, indicating a role for protective antibody-dependent and antibody-independent mechanisms of immunity. The involvement of gammadelta T cells in vaccine-induced PcAMA-1 and/or PcMSP-1(42) protection was minor. Analysis of the isotypic profile of antigen-specific antibodies induced by immunization of immunologically intact mice revealed a dominant IgG1 response. However, neither interleukin-4 and the production of IgG1 antibodies nor gamma interferon and the production of IgG2a/c antibodies were essential for PcAMA-1 and/or PcMSP-1(42) vaccine-induced protection. Therefore, for protective antibody-mediated immunity, vaccine adjuvants and delivery systems for AMA-1- and MSP-1-based vaccines can be selected for their ability to maximize responses irrespective of IgG isotype or any Th1 versus Th2 bias in the CD4(+)-T-cell response.
Figures
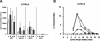

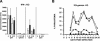

Similar articles
-
Immunization against Plasmodium chabaudi malaria using combined formulations of apical membrane antigen-1 and merozoite surface protein-1.Vaccine. 2003 May 16;21(17-18):1843-52. doi: 10.1016/s0264-410x(03)00026-4. Vaccine. 2003. PMID: 12706668
-
Prediction of merozoite surface protein 1 and apical membrane antigen 1 vaccine efficacies against Plasmodium chabaudi malaria based on prechallenge antibody responses.Clin Vaccine Immunol. 2009 Mar;16(3):293-302. doi: 10.1128/CVI.00230-08. Epub 2008 Dec 30. Clin Vaccine Immunol. 2009. PMID: 19116303 Free PMC article.
-
CD4+ T cells acting independently of antibody contribute to protective immunity to Plasmodium chabaudi infection after apical membrane antigen 1 immunization.J Immunol. 2000 Jul 1;165(1):389-96. doi: 10.4049/jimmunol.165.1.389. J Immunol. 2000. PMID: 10861076
-
Immunity to erythrocytic stages of malarial parasites.Am J Trop Med Hyg. 1994;50(4 Suppl):27-32. doi: 10.4269/ajtmh.1994.50.27. Am J Trop Med Hyg. 1994. PMID: 7909653 Review.
-
Immune responses to liver-stage parasites: implications for vaccine development.Chem Immunol. 2002;80:97-124. doi: 10.1159/000058841. Chem Immunol. 2002. PMID: 12058653 Review. No abstract available.
Cited by
-
Quantifying variation in the potential for antibody-mediated apparent competition among nine genotypes of the rodent malaria parasite Plasmodium chabaudi.Infect Genet Evol. 2013 Dec;20:270-5. doi: 10.1016/j.meegid.2013.09.013. Epub 2013 Sep 18. Infect Genet Evol. 2013. PMID: 24056014 Free PMC article.
-
Babesia divergens apical membrane antigen 1 and its interaction with the human red blood cell.Infect Immun. 2009 Nov;77(11):4783-93. doi: 10.1128/IAI.00969-08. Epub 2009 Aug 31. Infect Immun. 2009. PMID: 19720759 Free PMC article.
-
Enhanced protection against malaria by a chimeric merozoite surface protein vaccine.Infect Immun. 2007 Mar;75(3):1349-58. doi: 10.1128/IAI.01467-06. Epub 2006 Dec 11. Infect Immun. 2007. PMID: 17158895 Free PMC article.
-
Safety and immunogenicity of a malaria vaccine, Plasmodium falciparum AMA-1/MSP-1 chimeric protein formulated in montanide ISA 720 in healthy adults.PLoS One. 2008 Apr 9;3(4):e1952. doi: 10.1371/journal.pone.0001952. PLoS One. 2008. PMID: 18398475 Free PMC article. Clinical Trial.
-
Depletion and dysfunction of Vγ2Vδ2 T cells in HIV disease: mechanisms, impacts and therapeutic implications.Cell Mol Immunol. 2013 Jan;10(1):42-9. doi: 10.1038/cmi.2012.50. Epub 2012 Dec 17. Cell Mol Immunol. 2013. PMID: 23241900 Free PMC article. Review.
References
-
- Ahlborg, N., I. T. Ling, W. Howard, A. A. Holder, and E. M. Riley. 2002. Protective immune responses to the 42-kilodalton (kDa) region of Plasmodium yoelii merozoite surface protein 1 are induced by the C-terminal 19-kDa region but not the adjacent 33-kDa region. Infect. Immun. 70:820-825. - PMC - PubMed
-
- Anders, R. A., P. E. Crewther, S. Edwards, M. Margetts, M. L. S. M. Matthew, B. Pollock, and D. Pye. 1997. Immunization with recombinant AMA-1 protects mice against infection with Plasmodium chabaudi. Vaccine 16:240-247. - PubMed
-
- Batchelder, J. M., J. M. Burns, Jr., F. K. Cigel, H. Lieberg, D. D. Manning, B. J. Pepper, D. M. Yanez, H. van der Heyde, and W. P. Weidanz. 2003. Plasmodium chabaudi: IFN-γ but not IL-2 is essential for the expression of cell-mediated immunity against blood-stage parasites in mice. Exp. Parasitol. 105:159-166. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

