Alcohol tolerance in large-conductance, calcium-activated potassium channels of CNS terminals is intrinsic and includes two components: decreased ethanol potentiation and decreased channel density
- PMID: 15385615
- PMCID: PMC6729695
- DOI: 10.1523/JNEUROSCI.1536-04.2004
Alcohol tolerance in large-conductance, calcium-activated potassium channels of CNS terminals is intrinsic and includes two components: decreased ethanol potentiation and decreased channel density
Abstract
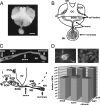
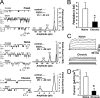
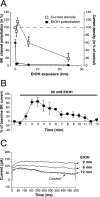
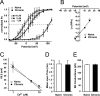
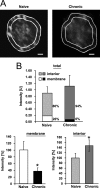
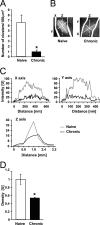
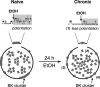
Tolerance is an important element of drug addiction and provides a model for understanding neuronal plasticity. The hypothalamic-neurohypophysial system (HNS) is an established preparation in which to study the actions of alcohol. Acute application of alcohol to the rat neurohypophysis potentiates large-conductance calcium-sensitive potassium channels (BK), contributing to inhibition of hormone secretion. A cultured HNS explant from adult rat was used to explore the molecular mechanisms of BK tolerance after prolonged alcohol exposure. Ethanol tolerance was intrinsic to the HNS and consisted of: (1) decreased BK potentiation by ethanol, complete within 12 min of exposure, and (2) decreased current density, which was not complete until 24 hr after exposure, indicating that the two components of tolerance represent distinct processes. Single-channel properties were not affected by chronic exposure, suggesting that decreased current density resulted from downregulation of functional channels in the membrane. Indeed, we observed decreased immunolabeling against the BK alpha-subunit on the surface of tolerant terminals. Analysis using confocal microscopy revealed a reduction of BK channel clustering, likely associated with the internalization of the channel.
Figures

Similar articles
-
Compartmentalized beta subunit distribution determines characteristics and ethanol sensitivity of somatic, dendritic, and terminal large-conductance calcium-activated potassium channels in the rat central nervous system.J Pharmacol Exp Ther. 2009 Jun;329(3):978-86. doi: 10.1124/jpet.108.146175. Epub 2009 Mar 25. J Pharmacol Exp Ther. 2009. PMID: 19321803 Free PMC article.
-
Molecular tolerance of voltage-gated calcium channels is evident after short exposures to alcohol in vasopressin-releasing nerve terminals.Alcohol Clin Exp Res. 2013 Jun;37(6):933-40. doi: 10.1111/acer.12057. Epub 2013 Feb 19. Alcohol Clin Exp Res. 2013. PMID: 23421938
-
Somatic localization of a specific large-conductance calcium-activated potassium channel subtype controls compartmentalized ethanol sensitivity in the nucleus accumbens.J Neurosci. 2004 Jul 21;24(29):6563-72. doi: 10.1523/JNEUROSCI.0684-04.2004. J Neurosci. 2004. PMID: 15269268 Free PMC article.
-
Ethanol interactions with calcium-dependent potassium channels.Alcohol Clin Exp Res. 2007 Oct;31(10):1625-32. doi: 10.1111/j.1530-0277.2007.00469.x. Alcohol Clin Exp Res. 2007. PMID: 17850640 Review.
-
Sizing up ethanol-induced plasticity: the role of small and large conductance calcium-activated potassium channels.Alcohol Clin Exp Res. 2009 Jul;33(7):1125-35. doi: 10.1111/j.1530-0277.2009.00936.x. Epub 2009 Apr 9. Alcohol Clin Exp Res. 2009. PMID: 19389201 Free PMC article. Review.
Cited by
-
A novel Ca(V)1.2 N terminus expressed in smooth muscle cells of resistance size arteries modifies channel regulation by auxiliary subunits.J Biol Chem. 2007 Oct 5;282(40):29211-21. doi: 10.1074/jbc.M610623200. Epub 2007 Aug 14. J Biol Chem. 2007. PMID: 17699517 Free PMC article.
-
A role for dynamin in triggering ethanol tolerance.Alcohol Clin Exp Res. 2012 Jan;36(1):24-34. doi: 10.1111/j.1530-0277.2011.01587.x. Epub 2011 Jul 28. Alcohol Clin Exp Res. 2012. PMID: 21797886 Free PMC article.
-
Posttranscriptional regulation of BK channel splice variant stability by miR-9 underlies neuroadaptation to alcohol.Neuron. 2008 Jul 31;59(2):274-87. doi: 10.1016/j.neuron.2008.05.032. Neuron. 2008. PMID: 18667155 Free PMC article.
-
An evolutionarily conserved mode of modulation of Shaw-like K⁺ channels.FASEB J. 2013 Apr;27(4):1381-93. doi: 10.1096/fj.12-222778. Epub 2012 Dec 11. FASEB J. 2013. PMID: 23233530 Free PMC article.
-
The molecular basis of tolerance.Alcohol Res Health. 2008;31(4):298-309. Alcohol Res Health. 2008. PMID: 23584007 Free PMC article. Review.
References
-
- Armstrong WE (1995) Morphological and electrophysiological classification of hypothalamic supraoptic neurons. Prog Neurobiol 47: 291–339. - PubMed
-
- Bonde C, Noraberg J, Zimmer J (2002) Nuclear shrinkage and other markers of neuronal cell death after oxygen-glucose deprivation in rat hippocampal slice cultures. Neurosci Lett 327: 49–52. - PubMed
-
- Brewer GJ (1997) Isolation and culture of adult rat hippocampal neurons. J Neurosci Methods 71: 143–155. - PubMed
-
- Burbach JP, Luckman SM, Murphy D, Gainer H (2001) Gene regulation in the magnocellular hypothalamo-neurohypophysial system. Physiol Rev 81: 1197–1267. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
