PIKE/nuclear PI 3-kinase signaling mediates the antiapoptotic actions of NGF in the nucleus
- PMID: 15385964
- PMCID: PMC524338
- DOI: 10.1038/sj.emboj.7600392
PIKE/nuclear PI 3-kinase signaling mediates the antiapoptotic actions of NGF in the nucleus
Abstract
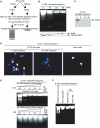
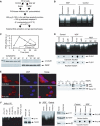
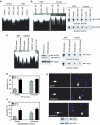
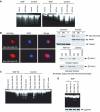
PI 3-kinase (PI3K) occurs in the nuclei of a broad range of cell types, and various stimuli elicit PI3K nuclear translocation. However, little is known about the biological function of nuclear PI3K. Here we show that nuclear PI3K and its upstream regulator PIKE mediate the antiapoptotic activity of nerve growth factor (NGF) in the isolated nuclei. The nuclei from NGF-treated PC12 cells, EGF-treated HEK293 cells and HeLa cells are resistant to DNA fragmentation initiated by activated cell-free apoptosome. Nuclei from constitutively active PI3K adenovirus-infected cells display the same resistance as those treated by NGF, whereas PI3K inhibitors, dominant-negative PI3K or PIKE abolishes it. Knockdown of either PI3K or PIKE diminishes the antiapoptotic activity of NGF. PI (3,4,5)P3 alone mimics the antiapoptotic activity of NGF, for which nuclear Akt is required. These results demonstrate that PIKE/nuclear PI3K signaling through nuclear PI (3,4,5)P3 and Akt plays an essential role in promoting cell survival.
Figures


References
-
- Andjelkovic M, Alessi DR, Meier R, Fernandez A, Lamb NJ, Frech M, Cron P, Cohen P, Lucocq JM, Hemmings BA (1997) Role of translocation in the activation and function of protein kinase B. J Biol Chem 272: 31515–31524 - PubMed
-
- Bao S, Tibbetts RS, Brumbaugh KM, Fang Y, Richardson DA, Ali A, Chen SM, Abraham RT, Wang XF (2001) ATR/ATM-mediated phosphorylation of human Rad17 is required for genotoxic stress responses. Nature 411: 969–974 - PubMed
-
- Bavelloni A, Santi S, Sirri A, Riccio M, Faenza I, Zini N, Cecchi S, Ferri A, Auron P, Maraldi NM, Marmiroli S (1999) Phosphatidylinositol 3-kinase translocation to the nucleus is induced by interleukin 1 and prevented by mutation of interleukin 1 receptor in human osteosarcoma Saos-2 cells. J Cell Sci 112: 631–640 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

